Based on their formulas which of these compounds is not likely to be an ionic compound: kbr, so2, or alcl3? explain.
Solved
Show answers
More tips
- S Style and Beauty How to knit a hooded cowl?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- H Health and Medicine How to perform artificial respiration?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- F Family and Home How to Properly Use a Water Level?...
Answers on questions: Chemistry
- M Mathematics will give ! the graph below shows the solution to a system of inequalities: which of the following inequalities is modeled by the graph? 4x − 3y ≤ 12; x ≥ 0 4x + 3y ≤ 12; x ≥ 0 4x...
- C Computers and Technology Someone please make a random question with an easy answer because i need one more brainliest to UPGRADE!...
- S Social Studies Help me please........................................
- B Biology If x stands for xylem and the p stands for phloem in the flax stem, then the flax plant is a...

Ответ:
Ответ:
The volume of hydrogen gas produced is 4.24 L
Explanation:
To calculate the number of moles, we use the equation:
Given mass of aluminium = 3.12 g
Molar mass of aluminium = 26.98 g/mol
Putting values in above equation, we get:
For the given chemical reaction:
As, NaOH is present in excess. It is considered as an excess reagent.
Aluminium is the limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
2 moles of aluminium produces 3 moles of hydrogen gas
So, 0.116 moles of aluminium will produce = of hydrogen gas
of hydrogen gas
To calculate the volume of hydrogen gas produced, we use the ideal gas equation:
where,
P = pressure of the gas = 750 torr
V = Volume of the gas = ?
T = Temperature of the gas =![20^oC=[20+273]K=293K](/tpl/images/0308/3868/3b5d4.png)
R = Gas constant =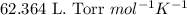
n = number of moles of the gas = 0.174 mol
Putting values in above equation, we get:
Hence, the volume of hydrogen gas produced is 4.24 L