golderhadashaowtatz
03.09.2019 •
Chemistry
Calculate ho298 for the process zn + s + 2o2 ? znso4 from the following information:
zn + s --> zns ho298 = -206.0kj
zns + 2o2 --> znso4 ho298 = -776.8 kj
Solved
Show answers
More tips
- L Leisure and Entertainment Learn How to Knit with Needles: A Simple Guide for Beginners...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
Answers on questions: Chemistry
- C Chemistry What is the pOH of a solution with H+ = 2.7 x 10^-2....
- C Chemistry Which is the best evidence that a chemical change has occurred? a. release of energy b. temperature change c. change of state d. formation of a new substance...
- C Chemistry Which gas is not known as green house gas...
- C Chemistry Which of the following elements is an alkali metal? a. potassium (k) b. calcium (ca) c. chromium (cr) d. germanium (ge)...
- C Chemistry Sodium reacts with chlorine gas according to the reaction: 2Na(s)+Cl2(g)→2NaCl(s) What volume of Cl2 gas, measured at 686 torr and 32 ∘C, is required to form 22 g of NaCl?...
- C Chemistry The main reason an author includes evidence in a case study is to help the reader create a case study. add to the case study. draw a conclusion from the case study. conduct an...
- C Chemistry PLEASE HELP!! I don t understand.....
- C Chemistry Determine the empirical formula of kevlar, used in making bullet proof vests, is 70.6% c, 4.2% h, 11.8% n and 13.4% o....
- C Chemistry Question 25 a chemist determined by measurements that 0.020 moles of mercury participated in a chemical reaction. calculate the mass of mercury that participated in the chemical...
- H History Which statement is untrue about Dr. Jonas Stalk? He developed a vaccine for polio by 1955. He patented the polio vaccine so people could only buy it from him. He helped create...

Ответ:
Explanation:
According to Hess’s law of constant heat summation, the heat absorbed or evolved in a given chemical equation is the same whether the process occurs in one step or several steps.
According to this law, the chemical equation can be treated as ordinary algebraic expression and can be added or subtracted to yield the required equation. That means the enthalpy change of the overall reaction is the sum of the enthalpy changes of the intermediate reactions.
The final reaction is:
By adding (1) and (2)
Hence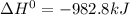 .
.
Ответ: