Tyrant4life
30.08.2019 •
Chemistry
Calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. it takes 4.2 joules to change 1.0 grm (1.0 ml) of water 1.0 celcius. is it endotherrmic or exothermic? volume of vinegar 25 ml, initial temp of vinegar 17 celcius and final temp is 14 celcius.
Solved
Show answers
More tips
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- B Biology 5. Cranberries have a pH between 2.3 and 2.5. This makes them an...
- M Mathematics -7 1/3 simplified help me...
- M Mathematics Sam is making a picture frame. He is gluing craft sticks end-to-end to a piece of cardboard. The sticks do not overlap. The top of the frame is 18 and one-third inches long. Each...

Ответ:
Explanation:
The given data is as follows.
Density of vinegar = 1.0 g/ml
Specific heat capacity = 4.25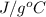
Relation between enthalpy and specific heat is as follows.
Hence, putting the values into the above formula as follows.
= (as density =
(as density = 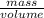 )
)
= - 315 J
Thus, we can conclude that the enthalpy of reaction is -315 J.
As the value is negative so, it means that heat is releasing. Hence, the reaction is exothermic in nature.
Ответ: