patrickdolano
18.03.2020 •
Chemistry
Calculate the mass of calcium phosphate and the mass of sodium chloride that could be formed when a solution containing 12.00g of sodium phosphate is added to a solution containing 10.0g of calcium chloride
Solved
Show answers
More tips
- F Food and Cooking How to Make Napoleon Cake: A Step-by-Step Guide...
- H Health and Medicine How to Increase Hemoglobin in the Blood...
- F Food and Cooking How to Make Chebureki: A Delicious Recipe for Russian Street Food...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- F Food and Cooking Is it Really Possible to Cook Tasty Colored Cauliflower?...
- H Health and Medicine Why do our Joints Crack?...
- H Health and Medicine These Food Additives Can Do Significant Harm to Your Health...
- F Food and Cooking How to Choose the Right Olive Oil: A Comprehensive Guide...
Answers on questions: Chemistry
- M Medicine What is an example of a mitochondrion...
- A Arts Help help pl (freepoints who up an wanna talk)...
- M Mathematics I need this done asap. What is the surface area for this net of a rectangular prism? (Only type in the number) pls no wrong answer....
- M Mathematics PLEASE HELP ‼️50 POINTS‼️...

Ответ:
Answer : The mass of calcium phosphate and the mass of sodium chloride that formed could be, 9.3 and 10.5 grams respectively.
Explanation : Given,
Mass of = 12.00 g
= 12.00 g
Mass of = 10.0 g
= 10.0 g
Molar mass of = 164 g/mol
= 164 g/mol
Molar mass of = 111 g/mol
= 111 g/mol
Molar mass of = 58.5 g/mol
= 58.5 g/mol
Molar mass of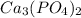 = 310 g/mol
= 310 g/mol
First we have to calculate the moles of and
and  .
.
and,
Now we have to calculate the limiting and excess reagent.
The balanced chemical reaction is:
From the balanced reaction we conclude that
As, 3 mole of react with 2 mole of
react with 2 mole of 
So, 0.0901 moles of react with
react with  moles of
moles of 
From this we conclude that, is an excess reagent because the given moles are greater than the required moles and
is an excess reagent because the given moles are greater than the required moles and  is a limiting reagent and it limits the formation of product.
is a limiting reagent and it limits the formation of product.
Now we have to calculate the moles of and
and 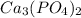
From the reaction, we conclude that
As, 3 mole of react to give 6 mole of
react to give 6 mole of 
So, 0.0901 mole of react to give
react to give 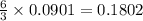 mole of
mole of 
and,
As, 3 mole of react to give 1 mole of
react to give 1 mole of 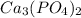
So, 0.0901 mole of react to give
react to give  mole of
mole of 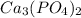
Now we have to calculate the mass of and
and 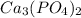
and,
Therefore, the mass of calcium phosphate and the mass of sodium chloride that formed could be, 9.3 and 10.5 grams respectively.
Ответ:
Cation and anion
Explanation:
Positive ion is known as cation and negative ion is known as anion