tytybruce2
25.02.2020 •
Chemistry
Calculate the percent ionization of propionic acid (C2H5COOH) in solutions of each of the following concentrations (Ka is given in Appendix D in the textbook).
Part A. 0.270 M
Part B. 7.84×10-2 M
Part C. 1.97×10-2 M
Solved
Show answers
More tips
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- P Physics What was the approximate average speed of the ocean current that carried the ducks to shore in m/s and in mi/h? (Rubber ducks from the same spill began to appear on the...
- E English What form of repetition is this - ah, but i know thou wilt do well in the battle, and thou shalt have all my prayers for thy safety and 15 success, said la belle isoude....
- H Health The final part of the small intestine, 10–12 feet long, is the a. duodenum. b. cecum. c. ileum. d. jejunum....
- M Mathematics 3/x^18= x to the th power...

Ответ:
a) = 0.704%
b) = 1.30%
c) = 2.60%
Explanation:
Given that:
For Part A; where Concentration of A = 0.270 M
Percentage Ionization(∝)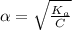
percentage% (∝) =
= 0.704%
For Part B; where Concentration of B =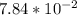 M
M
percentage% (∝) = 0.0130 × 100%
= 1.30%
For Part C; where Concentration of C=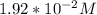
percentage% (∝) = 0.02608 × 100%
= 2.60%
Ответ:
Combination reaction