quise2ross
29.02.2020 •
Chemistry
Calculate the weight percent of ascorbic acid in a tablet of Vitamin C from the following data:A 80 mg sample of a crushed Vitamin C tablet was dissolved in 40 mL of H2SO4 and 20 mL of water. Two grams of KI and 40. mL of 0.00653 M KIO3 solution was added, and the mixture titrated to a starch endpoint. The titration required 15 mL of 0.0510 M thiosulfate solution. I know the answer is 88%, but how did they get it?
Solved
Show answers
More tips
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry Which statements correctly match the part of the microscope with its function? (choose all of the correct answers) A Ocular (Eyepiece) Lens: The knob that allows you...
- C Chemistry PLEASE HELP ASAP! AND PLEASE DO NOT GIVE ME ANY VIRUSES! A gardener plants a new kind of plant that will attract more helpful insects to his garden. Suddenly, the gardener...
- C Chemistry For the reaction represented by the equation H2O(g) + H2(g) + 1/2O2(g) the value of Delta G is...
- C Chemistry What is the valency of Ammonium Nitrite Nitrate Sulfite Sulfate Hydrogen Sulfate Hydroxide Phosphate Carbonate Hydrogen Carbonate Manganate (VII) Dichromate (VI)...
- C Chemistry 1 Determine the amount of heat (q) in joules that is released when 20.0 grams of water is cooled from 85˚C to 5˚C. Remember that the specific heat of water is 4.18 J/g˚C....
- C Chemistry Which table represents a relation that is not function? Please...
- C Chemistry 40.0 g metal at 34.3 ºC is placed in a calorimeter with 50.0 g water at 25.0 ºC. If the final temperature of the metal and water was 26.5 ºC, what is the specific heat...
- C Chemistry Explain the main idea of the aricle...
- M Mathematics Can you all ask dumb questions for yt please...
- M Mathematics 6.928 as a rationalized square root...

Ответ:
The Answer is 88%
Explanation:
The balanced ionic equation of the reaction
Looking at the above reactions
The original number of moles of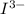 = 3 × number of moles of
= 3 × number of moles of 
= 3 × volume × concentration of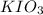
Note: The formula for number of moles is volume × concentration
The number of moles of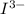 left after its reaction with ascorbic acid
left after its reaction with ascorbic acid
=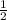 x moles of
x moles of 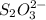
=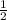 x volume x concentration of
x volume x concentration of 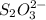
Note: The division by 1000 is to convert mill liter to liter
Moles of ascorbic acid = moles of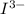 reacted
reacted
Hence
Mass of ascorbic acid = moles of ascorbic acid × molar mass of ascorbic acid
Weight% of ascorbic acid = mass of ascorbic acid/mass of sample x 100%
= 70.55/80 × 100%
= 88.1%
Ответ:
Electron is subatomic particles whose electric charge is negative one elementary charge (-1). Molecule who capture electrons now how more electrons than protons (one positive charge) and because of that that molecule is negative.