Can yall help me with this one, thanks!
Solved
Show answers
More tips
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry Citric acid is often used as an acidity regulator in hot water canning of tomatoes. at 25°c it has kat 7.4x10- and a.hº = +4.1 kl/mol for the acid dissociation process. what is...
- C Chemistry which property of metals is explained by the sea of electrons within metallic bonding? a. malleability b. all of the above c. electrical conductivity d. thermal conductivity...
- C Chemistry No show test 2117l semester 202012 pue (8/27) refer to the multiplication factor sheet to answer the following questions. write all answers in proper scientific form ie: if the...
- C Chemistry For the following reaction, 101 grams of magnesium nitride are allowed to react with 144 grams of water. mg3n2 (5) + 6 h20 (1) — 3 mg(oh)2 (aq) + 2 nh2 (aq) what is the formula...
- C Chemistry What is the thermodynamic equilibrium constant under standard conditions for the following balanced redox reaction? zr(s) + o2(g) - zro2 (s) een=2.463 v...
- C Chemistry Consider the following reactions. i. 2502(g) + o2(g) → 250,(g) over a pt(s) catalyst ii. 4hci(g) + o2(g) 2012(g) + 2h2o(g) over a cucl(s) catalyst iii. n2(g) + 3h2(g) + 2nh3(g)...
- C Chemistry what is the ph if 1ml of 0.1m hcl is added to 99ml of pure water? now if instead of pure water a buffer is used: hpo4-2/h2po4- pka = 7.2 assume the initial ph of this buffer is...
- C Chemistry The following kinetics data was collected at 826°c and at constant volume for the reaction: 2 no(g) + 2 h2(g) → n2(g) + 2 h2o(1) expt # pno, in bar ph,, in bar initial rate, in...
- C Chemistry Pre: 772/2900 convert 9.23 um to inches. given that: 1in = 2.54cm. 9.23 um = about us careen privacy policy terms of use...
- C Chemistry 4of 20 silver chloride (agcl) is relatively insoluble in water. at 25 c, 13x 10l of water is needed to dissolve 25g of aga what mass in milligrams) of agcl wil dissolve in 10 l...

Ответ:
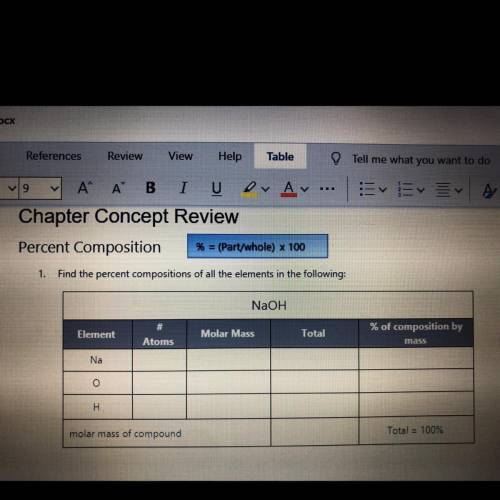
Element atom molar mass Total % composition
Na 1 23 40 57.5 %
O 1 16 40 40%
H 1 1 40 2.5 %
The given compound is NaOH.
Na : number of atom= 1
Molar mass= 23 g/mol
Total mass of NaOH = 40 g/mol
% composition by mass =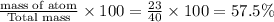
O : number of atom= 1
Molar mass= 16 g/mol
Total mass of NaOH = 40 g/mol
% composition by mass =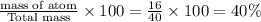
H : number of atom= 1
Molar mass= 1 g/mol
Total mass of NaOH = 40 g/mol
% composition by mass =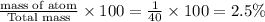
Ответ:
it would be vanquish