Cinética
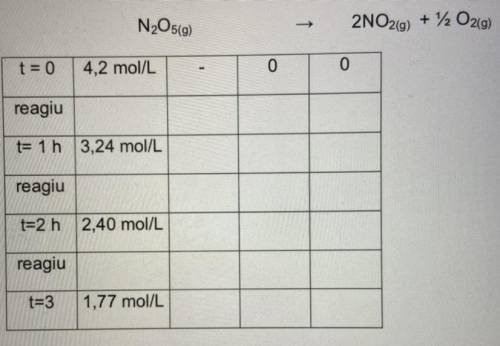
Considere a reação abaixo:
N2O5(g) → 2NO2(g) + 12 O2(g)
1. Inicialmente a concentração de N2O5 que esta dissolvido em tetracloreto
de carbono é 4,2 mol/L.
a) Qual a concentração inicial de NO2 e O2?
b) Após 1 h, ainda há N2O5 no recipiente e sua concentração é igual a
3,24mol/L. Calcule a velocidade média de decomposição da substância
no intervalo de 0 a 1 h.
c) Qual a concentração de N2O5, NO2 e O2 após 1 hora.
d) Após 2 h, a concentração de N2O5 e 2,40 mol/L. Calcule a velocidade
média de decomposição dessa substância no intervalo de 1 a 2 h.
e) Qual a concentração de N2O5, NO2 e O2 após 2 horas.
f) Após 3 h, a concentração de N2O5 é de 1,77. Calcule a velocidade média
de decomposição da substância no intervalo de 2 a 3 h.
g) Qual a concentração de N2O5, NO2 e O2 após 3 horas.
N205(9)
2NO2(g) + 12 O2(9)
Solved
Show answers
More tips
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry I’m in science class right now and I need help on these questions...
- H Health During which stage of an infectious disease does the body try to fight off pathogens before a person becomes ill? A) Recovery B)Incubation C)Inflammation D)Immunization...
- M Mathematics A basketball team plays 80% of its games at night. If it plays 32 games at night, what is the total number of games it plays?...
- M Mathematics A rectangular floor is 5yd long and 4yd wide. Amy wants to tile the floor with tile sold by the square foot. Use the facts to find the area in square feet....
- S Social Studies We are dead if we voted for either trump or biden (us) for LQTBQ+ community...
- C Computers and Technology . Which of the following is not advantages of computer network? * i. Data or program can be shared ii. All Hardware devices can be shared iii. Data processing is faster...

Ответ:
7.30g O₂ are in excess and 21.3g of SO₂ should be produced.
Explanation:
The reaction of Zinc sulfide (ZnS) with oxygen (O₂) produce ZnO and SO₂ as follows:
2ZnS + 3O₂ → 2ZnO + 2SO₂
Using this reaction we can find the amount of SO₂ produced converting the ZnS and O₂ to moles and finding limiting and excess reactant as follows:
Moles ZnS -Molar mass: 97.47g/mol-:
32.5g * (1mol / 97.47g) = 0.333 moles ZnS
Moles O₂ -Molar mass: 32g/mol-:
23.3g O₂ * (1mol / 32g) = 0.728 moles O₂
For a complete reaction of the 0.333 moles of ZnS are required:
0.333 moles ZnS * (3mol O₂ / 2mol ZnS) = 0.500 moles O₂.
As there are 0.728 moles of O₂, limiting reactant is ZnS and excess reactant is O₂.
The moles and mass of O₂ in excess are:
0.728mol - 0.500mol = 0.228moles O₂ * (32g / mol) = 7.30g O₂ are in excess
The produced mass of SO₂ -Obtained from the moles of ZnS- is:
0.333 moles of ZnS * (2mol SO₂ / 2mol ZnS) = 0.333 moles SO₂
0.333 moles SO₂ * (64.066g / mol) =
21.3g of SO₂ should be produced