jose0765678755
06.11.2020 •
Chemistry
Classify these reactions:
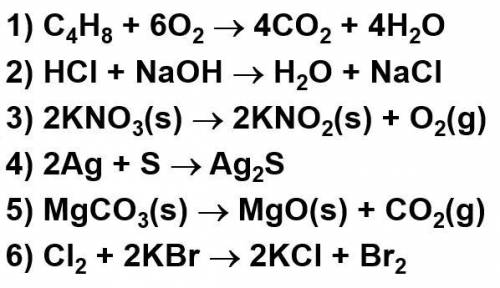
Solved
Show answers
More tips
- S Style and Beauty 5 Tips for Choosing the Best Ugg Boots...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- H Health and Medicine Effective Ways to Treat Colic in Infants...
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- C Cities and Countries What time does the Metro open in Moscow?...
- W Work and Career Where can you learn to be a flight attendant?...
- H Health and Medicine What are Electronic Cigarettes? The Ultimate Guide...
- C Computers and Internet Where are passwords stored in Opera?...
Answers on questions: Chemistry
- C Chemistry SOS HELP QUICK (explain if you can)...
- C Chemistry PLEASE HELP Elements in the same period are similar in that same number of valence electrons of same number of energy shells...
- C Chemistry An endothermic process is when energy (heat) is...
- C Chemistry Balance the equation C2H6O+O2 —— CO2 + H2O...
- C Chemistry The complete conversion of 90.0 grams of hydrogen to ammonia would require how many moles of nitrogen gas?...
- C Chemistry A compound contains oxygen and chlorine. Its molecular mass is 166.9g. What is the molecular formula if its empirical formula is CIO3?...
- C Chemistry Question #5: In the diagram of an eclipse below shadows are drawn in the 1 point Earth-Moon-Sun system and one side of Earth is labeled with an A and the opposite side...
- C Chemistry For the balanced equation shown below, if the reaction of 91.3 grams of C3H6 produces a 73.3% yield, how many grams of CO2 would be produced? 2C3H6+9O2= 6CO2+6H2O...
- C Chemistry What name is given to the elements in groups 3 through 12? how do their properties tend to compare with the elements to the left and right of these groups?...
- C Chemistry Sinkholes are often formed as a result of the actions...

Ответ:
-The principal quantum number refers to the main energy level.
-The angular momentum number refers to the type of orbital the electron is in.
-The magnetic quantum number refers to which orbital contains the electron.
-The spin quantum number distinguishes between the two electrons than any orbital can hold.
☃️ Hope this helps☃️