rosie20052019
14.09.2019 •
Chemistry
Combustion of 8.9541 grams of c4h10 will yield (example 2.345) do not enter units. grams of co2. (assume 100% yield)
Solved
Show answers
More tips
- S Science and Technology QR Code: How it Works and Why You Need it?...
- C Computers and Internet Dropbox: What is it and How to Use it...
- H Health and Medicine How to Increase Hemoglobin in the Blood...
- A Animals and plants How to Store Carrots: Tips for Homeowners...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- C Chemistry CaC2(s) + 2H2O(l) → C2H2(g) + Ca(OH)2(s). Suppose this reaction takes place in a sealed container in which the gas occupies a volume of 1.50 liters. The container...
- C Chemistry How does the corona virus related to chemistry?...
- C Chemistry One sugar produced by photosynthesis is called...
- C Chemistry You set up a radio transmitter that operates by moving electric charges through a wire in a north-south direction. If you turn the transmitter so that the charges...
- C Chemistry The amplitude of a wave is the measure of the...
- C Chemistry What is a common event due to transform plate movement at the San Andreas Fault Zone?...
- C Chemistry It takes blank for earth to rotate on its axis...
- C Chemistry Calculate the volume (in ml) of 0.170 m naoh that must be added to 389 ml of 0.0543 m 3-(n-morpholino)propanesulfonic acid (mops) to give the solution a ph of 7.55....
- C Chemistry Which of the following conditions remain constant in gay-lussac s law? temperature and number of moles volume and number of moles density and temperature volume and...
- B Business One day someone will love you the way you deserved to be loved and u won t have to fight for it...

Ответ:
The mass of carbon dioxide produced is 27.1 grams.
Explanation:
Combustion reaction is defined as the reaction in which a hydrocarbon reacts with oxygen gas to produce carbon dioxide gas and water molecule.
To calculate the number of moles, we use the equation:
Given mass of butane = 8.9541 g
Molar mass of butane = 58.12 g/mol
Putting values in equation 1, we get:
The chemical equation for the combustion of butane follows:
By Stoichiometry of the reaction:
2 moles of butane produces 8 moles of carbon dioxide
So, 0.154 moles of butane will produce =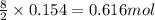 of carbon dioxide
of carbon dioxide
Now, calculating the mass of carbon dioxide by using equation 1, we get:
Moles of carbon dioxide = 0.616 moles
Mass of carbon dioxide = 44 g/mol
Putting values in equation 1, we get:
Hence, the mass of carbon dioxide produced is 27.1 grams.
Ответ: