Compare the contrast the arrangment of elements in mendeleev prodic table and mordern prodic table
Solved
Show answers
More tips
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
Answers on questions: Chemistry
- H History Change in caste system in later vedic period...
- C Computers and Technology Put the steps in order to produce the output shown below. Assume the indenting will be correct in the program. 52 53 82 83 1. Line 1 for numB in (5,8] 2. Line 2 for...
- H History When was the battle of Hastings? a. 1076b. 1090c. 1044e. 1066...
- S Spanish Match the word, country, or name with the correct phrase.match term definition poly glot a) these are ways of speaking that are rooted in latin.romance languagesb)...

Ответ:
The elements are arranged in the growing order of atomic masses in Mendeleev's periodic table. On the other hand, the cells are placed in the modern periodic table in the vastly increased order of increasing atomic numbers that is a more fundamental characteristic than the orbital radius.
Explanation:
The stance of the elements throughout the Mendeleev periodic table was to comply with the atomic mass. The position of the elements in the modern periodic table is controlled by the electronic structure, which evaluates their characteristics. The place of isotopes in Mendeleev's periodic table was not justified. The category is based mostly on the number of atoms and not the molar weight in the periodic table of elements and thus is justified the location of isotopes. The periodic table provided by Mendeleev had some linguistic factors on the atomic mass. There have been no defects in current periodic law as regards atomic number. For example, potassium had a lesser height in Mendeleev's periodic table than argon. But this has been solved in the Modern periodic table. Argon supersedes phosphorus as atomic number 18 is argon, so potassium has 19. The periodic table on Mendeleev does not authors would like to thank why the characteristics of the components are replicated at frequent intervals of 2, 8, 18, and 32. That is paper outlines by the periodic table of elements. The periodic table of Mendeleev contains no clear cut components of different types of elements. There is a clear cut separation of different types of elements in the modern periodic table, like representative elements, oxygen atoms, affect consumers. The periodic table provided by Mendeleev is not easy to remember. The periodic table Traditional is easy to recall.Ответ:
17.2 g
Explanation:
To calculate the moles :
The balanced chemical equation is:
According to stoichiometry :
4 moles of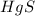 produce = 4 moles of
produce = 4 moles of 
Thus 0.085 moles of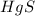 will require=
will require= of
of 
Mass of
Thus 17.2 g of will be produced form 20.0 g of HgS.
will be produced form 20.0 g of HgS.