codeyhatch142
20.05.2021 •
Chemistry
Consider two distinct aqueous solutions:
I - 0,75mol/L of C6H12O6
II - 0,3mol/L of Na2SO4.
Which of the two solutions will have the highest boiling temperature and why?
Solved
Show answers
More tips
- S Science and Technology Why is there no gravity on other planets?...
- F Food and Cooking Deflope: What is it and how does it work?...
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- F Family and Home How to Properly Use a Water Level?...
Answers on questions: Chemistry
- C Chemistry What type of rock forms from thin and runny, fast-moving lava?...
- E English How does the mood of the opening scene contrast with the mood at the start of Scene 2 ? The Tempest...
- B Biology What does a phenotype ratio of 3:1 among offspring in a monohybrid cross indicate?...
- S Social Studies The issue of being extended into new territories was becoming a crisis....
- A Arts Take me to the dark faling to the finish the liric and tell me the aname of the artist and ill give you brainlist...
- M Mathematics Bo and Erica are yoga instructors. Between the two of them, they teach 39 yoga classes each week. If Erica teaches 15 fewer than twice as many as Bo, how many classes does...

Ответ:
The rate law for the given reaction is![\boxed{rate=k\left[ {{{\text{X}}_2}}\right]\left[ {\text{Y}} \right]}](/tpl/images/0127/2448/0a486.png) .
.
Further explanation:
The rate of reaction is defined decrease in concentrations of reaction in unit time or increase in products concentration in unit time. Rate of reaction is determined by the rate law. The rate law is a relationship between rate constant and reactants of the reaction.
Consider a reaction,
The rate law for the general equation is expressed as,
Here,
rate is the rate of the reaction.
k is the rate constant.
[A] and [B] are the concentration of reactant species.
a is the order of reaction with respect to reactant A.
b is the order of reaction with respect to reactant A.
The given reaction is,
The rate law for this reaction is can be written as follows:
Here, a, b, and c are order of reaction with respective to reactant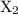 , Y, and Z respectively.
, Y, and Z respectively.
Since it is found that rate of reaction is doubled on doubling the concentration of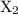 thus the order of reaction with respect to the
thus the order of reaction with respect to the 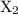 is 1, and therefore, the value of a is 1.
is 1, and therefore, the value of a is 1.
The rate of reaction is doubled on doubling the concentration of Y thus, order of reaction with respect to reactant Y is 1, and therefore, the value of b is also 1.
The rate of reaction does not affect on doubling the concentration of reactant Z thus order of reaction is not depended on the reactant Z, and therefore, the value of c is 0.
Substitute these values in equation (2).
Learn more:
1. Calculation of equilibrium constant of pure water at 25°C:
2. Complete equation for the dissociation of :
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Kinetics
Keywords: Kinetics, rate of reaction, x2+y+z->xy+xz, rate=kX2Y, zero order, first order, second, order.