xoxolovezzz7181
09.07.2019 •
Chemistry
Determine the mass percent of each of the elements in the fungicide copper(ii) oleate, cu(c18h33o2)2.
Solved
Show answers
More tips
- H Health and Medicine Contrast Shower: Benefits for the Body and Soul...
- S Society and Politics Skoptsy: Who They Are and How They Perform Castration?...
- H Health and Medicine How to Calculate Your Ideal Weight?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry Which of the following is true of a solid? A. The particles of a solid vibrate around a fixed point. B. The particles of a solid flow around each other within a fixed volume....
- C Chemistry Given the relative abundance of the following naturally occurring isotopes of oxygen, calculate the average atomic mass of oxygen: oxygen-16: 99.76% oxygen-17: 0.037% oxygen-18:...
- C Chemistry If you had a sample containing 55 alpha particles, how many protons would be present in that sample? show all necessary work....
- C Chemistry Define ionization energy and explain why the second aisle and ionization energy of the element is higher than its ionization energy...
- C Chemistry 2H2 (g) + O2 (g) → 2H20 (1) How many grams of oxygen gas is needed to create 160 g H20?...
- C Chemistry Does anyone knows how to solve that? I m kinda stuck there T_T...
- C Chemistry What is the mole fraction of carbon dioxide in a mixture consisting of equal masses of CO2 (MW=44) and neon (MW=20.2)?...
- C Chemistry Pleeeaassseeee I need help...
- C Chemistry An unknown metal M reacts with S to form a compound with a formula M₂S₃ . If 3.12g of M reacts with exactly 2.88 g of S, what is the name of metal M and compound M₂S₃ ? 2M +3S...
- C Chemistry The molecule (CH3)2N-PF2 has two basic atoms P and N . One is bound to B in a complex with BF3, the other to B in a complex with BF3 . Decide which is which and state your reason?...

Ответ:
The mass percent is determined by the formula:
mass percent =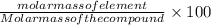 -(1)
-(1)
Given compound is copper(II) oleate,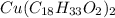
The atomic mass of the elements present in copper(II) oleate,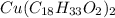 are:
are:
Atomic mass of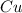 =
= 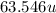
Atomic mass of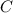 =
= 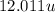
Atomic mass of =
= 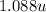
Atomic mass of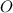 =
= 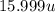
The molar mass of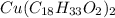 =
= 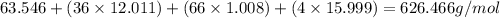
Using formula (1), the mass percent of each element is calculated as:
ForMass percent of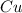 =
= 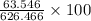 %
%
Mass percent of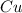 =
= 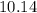 %.
%.
ForMass percent of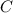 =
= 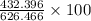 %
%
Mass percent of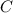 =
= 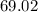 %.
%.
ForMass percent of =
= 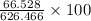 %
%
Mass percent of =
= 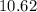 %.
%.
ForMass percent of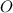 =
= 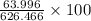 %
%
Mass percent of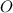 =
= 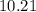 %.
%.
Ответ:
Answer and Explanation:
There are nine atoms in a molecule of ethanol.
Explanation:
The chemical formula can determine the number of atoms in a molecule.