cristianc52
17.05.2021 •
Chemistry
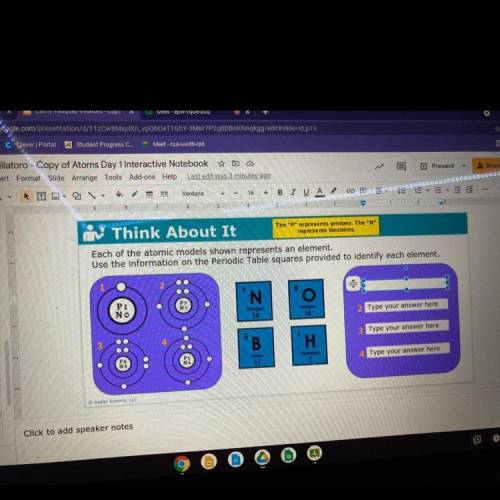
Each of the atomic models shown represents an element.
Use the information on the Periodic Table squares provided to identify each element.
8
Pi
NO
N
Nitrogen
14
Oxygen
16
2 Type your answer here
5
B
Type your answer here
B
H
Boron
11
Hydrogen
1
4. Type your answer here
Solved
Show answers
More tips
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- P Philosophy How to Develop Extrasensory Abilities?...
Answers on questions: Chemistry
- C Chemistry What is the order of the phases on a heating curve...
- C Chemistry Me! the symbol sb stands for stibnum or stibnite. what is the modern name of this element?...
- C Chemistry How many protons electrons and neutrons does 81br have?...
- C Chemistry Nic bought 12 pens for 2$ each and 12pads of paper for 3$ each. write two expressions for how nic could calculate his total.write one expression in expanded form and one in factored...
- C Chemistry An atom has atomic number of 34. how many protons and electrons does it have? what is its symbol? 34 protons and electrons. se list the number of...
- C Chemistry Meet url:-qxh-wtzk-xekSex meeting...
- C Chemistry The diagram shows copies of original waves. based on the data which statement describes the most reliable way to encode and transmit data and tells why? -please help ASAP + offering...
- C Chemistry How many grams of koh would be needed to produce 5.00 moles of k2so4?...
- C Chemistry Is a solids shape fixed, or can it be changed?...
- C Chemistry a gas occupies 1.85 L at 819 torr and 15*C What volume will it fill if the pressure is changed to 712 torr and the temperature is raised to 46*C (in L)...

Ответ:
CH₄ + 2 Cl₂ → CCl₄ + 4 HCl
According to Balanced Equation,
44.8 L (2 moles) Cl₂ gas when consumed gives = 22.4 (1 mole) L CCl₄ gas
So, for 1.1 mL (0.001 L) Cl₂ gas, the amount of CCl₄ produced will be,
0.001 L Cl₂ gas on consumption will give = X L of CCl₄ gas
Solving for X,
X = (0.001 L × 22.4 L) ÷ 44.8 L
X = 0.005 L of Cl₄
Or,
X = 5 mL of CCl₄
Result:
On consumption of 1.1 mL of Cl₂ gas, 5 mL of CCl₄ gas is produced.