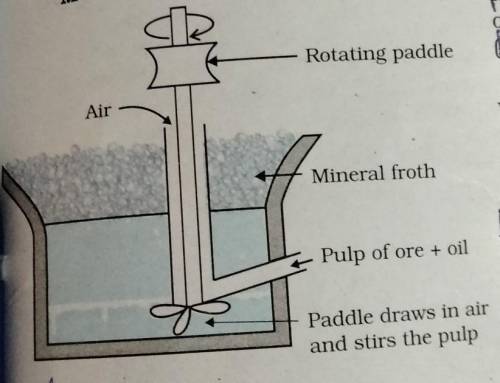
Explain this method (Froth floatation method)..........
Solved
Show answers
More tips
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...

Ответ:
froth flotation is a technique commonly used in the mining industry. In this technique, particles of interest are physically separated from a liquid phase as a result of differences in the ability of air bubbles to selectively adhere to the surface of the particles, based upon their hydrophobicity.
Explanation:
Froth floatation method is commonly used to concentrate sulphide ore such as galena (PbS), zinc blende (ZnS) etc. (ii) In this method, the metaalic ore particles which are perferentially wetted by oil can be separated from gangue. (iii) In this method, the crushed ore is suspended in water and mixed with frothing agent such as pine oil, eucalyptus oil etc. (iv) A small quantity of sodium ethyl xanthate which act as a collector is also added. (v) A froth is generated by blowing air through this mixture. (vi) The collector molecules attach to the ore particles and make them water repellent. (vii) As a result, ore parrticles, wetted by the oil, rise to the surface along with the froth. (viii) The froth is skimmed off and dried to recover the concentration ore. (ix) The gangue particles that are preferentially wetted by water settle at the bottom.
Ответ:
1. I hypothesize that if you continue to add more drops, then the time it takes will continue to increase.
2. A; 9.70 secs, B; 14.63 secs, C; 18.78 secs, D; 28.70 secs, E; 31.45 secs.
3. The formation continued to increased. As more was added, the time it took to reach the full reaction was increased.
4. Yes.
5. When the concentration of all the reactants increases, more molecules or ions interact to form new compounds, and the rate of reaction increases. When the concentration of a reactant decreases, there are fewer of that molecule or ion present, and the rate of reaction decreases.
6. 6mL.
Explanation:
for number one remember that the hypothesis doesnt have to be long or right, so you can basically put what you like if you truly dont know. but in this experiment they are asking you what you think will happen to the length of time. for number 2, i measured the time of when each reaction turned black.in the video, as more of the drops were added it took longer for it to turn black,, which is why i worded my answer that way. they are just asking did the time increase as you added more except this time they want the correct answer.in this case my hypothesis was correct,, but you can change your hypothesis and put no for 4 if it makes you feel better.i honestly looked this one up. 6 is correct but i hope this helps.