deepspy599otchpd
27.07.2020 •
Chemistry
Find the percentage composition of each element in the compound having 9.8 grams of nitrogen,0.7 grams of hydrogen and 33.6 grams of oxygen
Solved
Show answers
More tips
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- H Health and Medicine How to perform artificial respiration?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Properly Use a Water Level?...
Answers on questions: Chemistry
- E English analyz the two texts to determine which writers presents the stronger case?Speech: Proposal for Automatic Traffic Light Cameras by Councilman Lorenzo Hart Baler Town Council meeting...
- M Mathematics The sum of twice a number and 13 is 47. Find the number....
- H Health Only certain sports rely on a special set of muscles. A. True B. FalseI will report u if you put fake answer...
- C Chemistry An element has three naturally occurring isotopes with atomic masses of 25.958 amu, 27.242 amu, and 29.670 amu. The relative abundances of these elements are 92.2%, 4.69%, and 3.08%,...

Ответ:
The percentage composition of nitrogen , hydrogen and oxygen is 22.2 % , 1.59 % and 76.2% respectively.
Explanation:
Percentage composition is defined as the ratio of mass of substance to the total mass in terms of percentage.
Percentage composition=
a)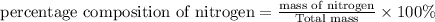
b)
c)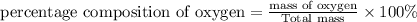
The percentage composition of nitrogen , hydrogen and oxygen is 22.2 % , 1.59 % and 76.2% respectively.
Ответ:
Heinrich Heine wrote in his 1820-1821 play Almansor the famous admonition, "Dort, wo man Bücher verbrennt, verbrennt man am Ende auch Menschen": "Where they burn books, they will in the end also burn people."
Explanation: