Four samples of gas each exert 675 mm hg in separate 3.5 l containers. what pressure will they exert if they are all placed in a single 3.5 l container? a.169 mm hg b.675 mm hg c.2.36 x 103 d. 2.70 x 103
Solved
Show answers
More tips
- S Science and Technology QR Code: How it Works and Why You Need it?...
- C Computers and Internet Dropbox: What is it and How to Use it...
- H Health and Medicine How to Increase Hemoglobin in the Blood...
- A Animals and plants How to Store Carrots: Tips for Homeowners...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- C Chemistry A 40.0L tank of ammonia has a pressure of 12.7kPa. Calculate the volume of the ammonia if its pressure is changed to 8.4 kPa while its temperature remains constant...
- C Chemistry How do surface currents differ from deep ocean currents? Deepwater currents are dependent on the wind and landmasses. Deepwater currents are dependent on temperature and...
- C Chemistry Ture or False: air has its components spread evenly, so it is considered a homogenous mixture....
- C Chemistry Can someone help me ????...
- C Chemistry Which object contains molecules with the slowest movement? a.cup of hot coffee b.helium balloon c.glass of water d.science book...
- C Chemistry Help me i mark brain thing...
- C Chemistry What is the total pressure in atmof a mixture of three gases with partial pressures of 11.5kPa, 30.6 kPa and 12.2 kPa?...
- C Chemistry How many mole are in 14.5 grams of neon please explain...
- C Chemistry What are the effects on people when mosquito fail to reproduce and become extinct...
- C Chemistry For the following balanced reaction, calculate how many moles of product would be produced by the complete conversion of 0.15 mol of magnesium 2Mg(s) + 03(g) --- 2MgO(s)...

Ответ:
if each gas occupies 675 mmhg
what about 4 gases in the sample
by cross multiplication
= 675 mm hg x 4/1 = 2.7 x10^3mmhg (answer D)
Ответ:
The question is incomplete, the complete question is:
The solubility of slaked lime, , in water is 0.185 g/100 ml. You will need to calculate the volume of
, in water is 0.185 g/100 ml. You will need to calculate the volume of  M HCl needed to neutralize 14.5 mL of a saturated
M HCl needed to neutralize 14.5 mL of a saturated
The volume of HCl required is 290mL, the mass of is 0.0268g, the moles of
is 0.0268g, the moles of
Explanation:
Given values:
Solubility of = 0.185 g/100 mL
= 0.185 g/100 mL
Volume of = 14.5 mL
= 14.5 mL
Using unitary method:
In 100 mL, the mass of present is 0.185 g
present is 0.185 g
So, in 14.5mL. the mass of present will be =
present will be =
The number of moles is defined as the ratio of the mass of a substance to its molar mass.
The equation used is:
Given mass of = 0.0268 g
= 0.0268 g
Molar mass of = 74 g/mol
= 74 g/mol
Plugging values in equation 1:
Moles of present =
present = 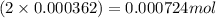
The chemical equation for the neutralization of calcium hydroxide and HCl follows:
By the stoichiometry of the reaction:
Moles of = Moles of
= Moles of  = 0.000724 mol
= 0.000724 mol
The formula used to calculate molarity:
Moles of HCl = 0.000724 mol
Molarity of HCl =
Putting values in equation 2, we get:
Hence, the volume of HCl required is 290mL, the mass of is 0.0268g, the moles of
is 0.0268g, the moles of