mrnotsosmart744
05.07.2020 •
Chemistry
g Which statement is incorrect regarding oxidation? Oxidation is a "gain" of electrons. Oxidation is the combination with O atoms. Oxidation is an increase in oxidation state. Oxidation is always accompanied by reduction. none of these
Solved
Show answers
More tips
- G Goods and services Should You Engrave Your Wedding Rings? Facts, Myths, and Considerations...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry List the three atomic particles, their relative masses and their, charges. why is the mass of an electron ignored when calculating the mass of an atom?...
- C Chemistry How many moles are in a pill containing 37.0mg of c12h22feo14...
- C Chemistry Ne(g) effuses at a rate that is times that of xe(g) under the same conditions....
- C Chemistry What is the perfect composition of calcium in calcium chloride?...
- C Chemistry What is the new concentration of a solution of CaSO3 if 10.0 mL of a 2.0 M CaSO3 solution is diluted to 100 ml?...
- C Chemistry The dark areas on the world map show places with climates that are alike. The table shows information for those places. A student is using the map and table to describe the climates...
- C Chemistry Summarize the process of the “stormy voyage to the island of stability” described at the end of the article....
- C Chemistry in the dehydration experiment to be performed this week, after acid is added to the alcohol, what is the general name of the first reaction intermediate that is formed?...
- E Engineering A garment to protect the wearer from toxic agents may be made of a fabric that cantains an adsorbent such as activated carbon. in a test of such a fabric, a gas stream containing...
- C Chemistry You have a metal with mass 15.32 g and a volumetric flask with water with mass 115.16 g. The mass of the volumetric flask with water and the metal is now 128.77 g. The empty volumetric...

Ответ:
The incorrect statement from the options is OXIDATION IS A "GAIN" OF ELECTRONS
Explanation:
Oxidation in a redox reaction is the loss of electrons. It is also the increase in the oxidation states of an atom or ion or atoms in a molecule. A redox reaction is a type of chemical reaction in which there is a transfer of electrons from an atom or ion to another resulting in a change in oxidation states of the substances involved. The reducing agent in the reaction is undergoes oxidation by losing electrons while the oxidating agent is reduced that is it gains electrons at the end of the reaction. The atom or ion from which electron is lost is said to be oxidized while the other atom or ion involved in the reaction is reduced.
Oxidation is also the combination with O atoms and it is always accompanied by reduction because oxidation forms a half of the whole redox reaction. A substance cannot be oxidized except it has reduced another substance by losing electrons to it.
Ответ:
Step-by-step explanation:
Using tangent ratio:
As per the statement:
In the given right angle triangle.
First find x:
Using tangent ratio:
⇒x = 7 units
We have to find the value of y:
Using Pythagoras theorem;
Substitute the given values we have;
⇒
⇒
⇒ units
units
Therefore, the value of y is,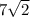 units
units