kathleen1010
06.12.2019 •
Chemistry
How can the solubility of alcohols be estimated?
Solved
Show answers
More tips
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
Answers on questions: Chemistry
- C Chemistry En el laboratorio se requiere preparar medio litro de una solución 1.5 molar de sulfato de sodio. a. ¿Cuántos gramos de sulfato de sodio se necesitan? b. ¿Cuántos mL de...
- C Chemistry Helpppppppppp pleaseeeeeee...
- C Chemistry Fill in the blanks: a substance that undergoes a change will always result in something new that has properties than what it started out as. A- physical and different. b-...
- C Chemistry 2. What is the purpose of subscripts? 3. What is the difference between monatomic and polyatomic ions? 4. How do the names of monatomic anions end? 5. When are Roman numerals...
- C Chemistry I have to have this done by 1:00 help meeeeee...
- C Chemistry A person standing 35 feet from a street light casts a shadow, as shown. What is the height h of the street light?...
- B Biology Compare the chemical equation for photosynthesis and cellular respiration.Explain how the two processes are interrelated....
- M Mathematics X-intercept:3 y-intercept:3...
- C Chemistry Calculate the mass of SO2, needed to produce 530 g of SO3....
- M Mathematics Need Help Isabella spots an airplane on radar that is currently approaching in a straight line, and that will fly directly overhead. The plane maintains a constant altitude...

Ответ:
Ответ:
44.04 g/mol
Explanation:
Ideal gas law in terms of density:
where,
P = pressure of the gas = 0.789 atm
V = volume of the gas
T = temperature of the gas =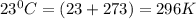
n = number of moles of the gas
R = gas constant = 0.0821 Latm/moleK
M = molar mass of gas
w = mass of gas
Thus molar mass of the gas is 44.04 g/mol