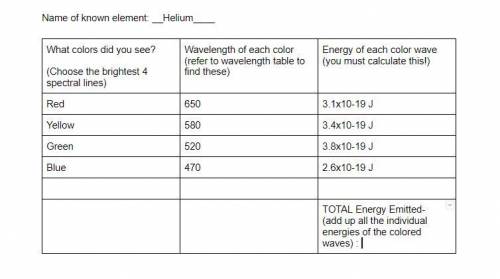
How do I add up all the energy to get the total energy emitted?
Solved
Show answers
More tips
- S Science and Technology Why is there no gravity on other planets?...
- F Food and Cooking Deflope: What is it and how does it work?...
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- F Family and Home How to Properly Use a Water Level?...
Answers on questions: Chemistry
- C Chemistry The hydroxyl radical, a fragment of water vapor known as the atmospheric cleanser , with a chemical formula of OH, has a globally averaged number density of ~1×10^6...
- C Chemistry Question 5 IPL What pressure is required to contain 0.034 moles of oxygen gas in a 5.1 L container at a temperature of 25.0°C? (use the R value for atm) O O 0.16 atm...
- C Chemistry A solution of the ionic salt NaCl would have pH....
- C Chemistry If a reaction vessel initially contains an N2O4N2O4 concentration of 0.0550 MM at 500 KK , what are the equilibrium concentrations of N2O4N2O4 and NO2NO2 at 500 KK...
- C Chemistry a rigid ylinder with a movable piston ontains 50.0 liters of a gass at 30.0 C with a pressure of 1.00 atmosphere, what is the volume of the gas in the cylinder at STP...
- C Chemistry Label each of the following measurements by the quantity each represents. For instance, a measurement of 10.6kg/m3 represents density. Measurement. Quantity the measurement...
- C Chemistry What is the wavelength of a 2.34*10^9/s freaquency...
- C Chemistry What is the mass number of a carbon isotope that has seven neutrons...
- C Chemistry Identify the strongest acid from the list below: КОН HNO3 HCOOH HF...
- C Chemistry PLEASE HELP ITS MY FINAL 1. The number of moles in 9 grams of aluminum is 3. TRUE or FALSE...

Ответ:
To calculate the amount of heat released in a chemical reaction, use the equation Q = mc ΔT, where Q is the heat energy transferred (in joules), m is the mass of the liquid being heated (in kilograms), c is the specific heat capacity of the liquid (joule per kilogram degrees Celsius).
Explanation:
Ответ: