How do I perform a titration on coca-cola or Pepsi, when I cannot see the color change because the color is too dark. The indicator is phenolphthalein.
Solved
Show answers
More tips
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
Answers on questions: Chemistry
- S Social Studies What does Loftus mean when she states that, Memory is like a Wikipedia page? Describe in good detail two examples given by Loftus that demonstrate how misinformation can create false...
- M Mathematics A password is required to be 12 to 16 characters in length. Characters can be digits (0-9), upper or lower-case letters (A-Z, a-z) or special characters. There are 10 permitted special...
- S Spanish Activty 21 un sabado con la famila...

Ответ:
maybe you need to do very dilute solution?
Explanation: you should have an pH meter instead of indicator.
Phosphoric acid H3PO4 have three ekvivalent points.
Another way could be using a conductivity meter.
Ответ:
HBr has the lowest rate of effusion at a given temperature.
Explanation: The effusion rate usually increases with increase in temperature because the kinetic energy of the gaseous molecules increases. But it was not true for gases having heavier mass. This was explained by Graham's Law.
Graham's Law states that the rate of effusion of a gas is inversely proportional to the square root of its molecular weight.
We are given different gases with different Molecular masses. The gas having larger Molecular mass will have the lowest rate of effusion.
Mol. Mass of = 80 g/mol
= 80 g/mol
Mol. Mass of = 16 g/mol
= 16 g/mol
Mol. Mass of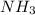 = 17 g/mol
= 17 g/mol
Mol. Mass of HBr = 81 g/mol
Mol. Mass of HCl = 36 g/mol
As, Mol. mass of HBr is the highest, so its rate of effusion will be the lowest.