reghanhoward3
08.10.2019 •
Chemistry
How many atoms of gold are in 112 grams of gold?
Solved
Show answers
More tips
- A Animals and plants Money Tree Care Secrets: How to Keep Your Plant Thriving...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- P Philosophy How to Develop Extrasensory Abilities?...
- S Style and Beauty Don t Sacrifice Your Brows: How to Properly Pluck Stubborn Hairs...
Answers on questions: Chemistry
- C Chemistry What is the name for As4010?...
- C Chemistry PLEASE HELP ASAP How many moles are there in 2.25 x 10^25 atoms of Zinc? How many atoms are there in 9500.0 mg of Fluorine?...
- C Chemistry Pleasssseeeee hellppppp nowwww...
- C Chemistry During STATIC ELECTRICITY to what type of objects & direction do electrons ALWAYS move to?...
- C Chemistry Atomic Structure o N ACROSS 4 The dense center of the atom where protons and neutrons are located E 5 U The area that surrounds the nucleus: Where negative subatomic...
- C Chemistry Whats the definition of collision?...
- M Mathematics Find an nth degree polynomial function with real coefficients satisfying the given conditions. degree of the polynomial is 3; the zeros of the polynomial are 3 and...
- H History Write the statement of the problem 3 or more questions...
- M Mathematics Find the equation of the line that passes through (14,-9) with slope 0...
- C Chemistry 2) The pH of 1x10-1M (HNO3) solution is: A)1 B) 1.3 C)5 D)2...

Ответ:
196.96 g 6.02x10²³ atoms
112 g ( atoms)
atoms = 112 x ( 6.02x10²³) / 196.96
atoms = 6.74x10²⁵ / 196.96
= 3.42x10²³ atoms
hope this helps!
Ответ:
The pH of resulting solution is 9.08
Explanation:
To calculate the number of moles for given molarity, we use the equation:
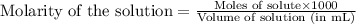 ........(1)
........(1)
For HCl:Molarity of HCl = 0.40 M
Volume of solution = 15.0 mL
Putting values in equation 1, we get:
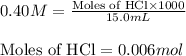
For ammonia:Molarity of ammonia = 0.50 M
Volume of solution = 20.0 mL
Putting values in equation 1, we get:
The chemical reaction for hydrochloric acid and ammonia follows the equation:
Initial: 0.006 0.01
Final: - 0.004 0.006
Volume of solution = 15.0 + 20.0 = 35.0 mL = 0.035 L (Conversion factor: 1 L = 1000 mL)
To calculate the pOH of basic buffer, we use the equation given by Henderson Hasselbalch:We are given:
pOH = ?
Putting values in above equation, we get:
To calculate pH of the solution, we use the equation:
Hence, the pH of the solution is 9.08