macylen3900
12.12.2019 •
Chemistry
How many ml of 0.100 m naoh are needed to neutralize 50.00 ml of a 0.150 m solution of ch3co2h, a monoprotic acid? how many ml of 0.100 m naoh are needed to neutralize 50.00 ml of a 0.150 m solution of ch3co2h, a monoprotic acid?
a. 37.50 ml
b. 50.00 ml
c. 75.00 ml
d. 100.00 ml
e. 25.00 ml
Solved
Show answers
More tips
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- E English 40 points ok so i have to write a memoir of 500-750 words about a personal experience that is important for the actions i chose and consequences of your actions or...
- H History Why is AC called “alternating current”?...
- M Mathematics Why did minnie die in the book ten...
- M Mathematics Using the given equation, (a) find the intercepts of its graph and (b) use the intercepts to graph the equation. (a) Select the correct choice below and, if necessary,...

Ответ:
We need 75 mL of 0.1 M NaOH ( Option C)
Explanation:
Step 1: Data given
Molarity of NaOH solution = 0.100 M
volume of 0.150 M CH3COOH = 50.00 mL = 0.05 L
Step 2: The balanced equation
CH3COOH + NaOH → CH3COONa + H2O
Step 3: Calculate moles of CH3COOH
Moles CH3COOH = Molarity * volume
Moles CH3COOH = 0.150 M * 0.05 L
Moles CH3COOH = 0.0075 moles
Step 4: Calculate moles of NaOH
For 1 mol of CH3COOH we need 1 mol of NaOH
For 0.0075 mol CH3COOH we need 0.0075 mole of NaOH
Step 5: Calculate volume of NaOH
volume = moles / molarity
volume = 0.0075 moles / 0.100 M
Volume = 0.075 L = 75 mL
We need 75 mL of 0.1 M NaOH
Ответ:
Answer :
The specific heat of calorimeter is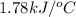
The energy of combustion per mole of vanillin is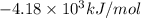
Explanation :
Part 1 :
First we have to calculate the energy released for 0.1625 g of benzoic acid.
Energy released = Energy released × Mass of benzoic acid
Energy released = (26.42 kJ/g) × (0.1625g)
Energy released = -4.293 kJ
Now we have to calculate the specific heat of calorimeter.
Heat released by the reaction = Heat absorbed by the calorimeter
where,
Now put all the given values in the above formula, we get:
Thus, the specific heat of calorimeter is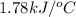
Part 2 :
First we have to calculate the energy released by the reaction.
where,
Now put all the given values in the above formula, we get:
Now we have to calculate the energy of combustion per mole of vanillin.
Molar mass of vanillin = 152.15 g/mole
Mass of vanillin = 0.2070 g
Thus, the energy of combustion per mole of vanillin is