How many moles of nano3 are present in a 44.0 g sample
Solved
Show answers
More tips
- P Philosophy Agnosticism: Opinion or Belief?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- M Mathematics What is 24288 rounded off to the nearest thousand...
- M Mathematics A rocket travels according to the equation h = - 4.9(t - 6)^2 + 182, How high was the rocket above the ground when it was fired?...
- S Social Studies How does the poem Evangeline describe the plight of the Acadian people? PLZZ HELP! 3...
- M Mathematics The difference between a number and 12 is 20...
- H Health Hallucinogens distort an individual s perceptions of reality. Please select the best answer from the choices provided. T F...

Ответ:
Answer : The number of moles of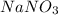 present in a given sample are 0.518 moles.
present in a given sample are 0.518 moles.
Explanation : Given,
Mass of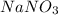 = 44.0 g
= 44.0 g
Molar mass of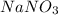 = 84.99 g
= 84.99 g
Formula used :
Now put all the given values in this formula, we get:
Therefore, the number of moles of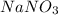 present in a given sample are 0.518 moles.
present in a given sample are 0.518 moles.
Ответ:
hint: formula
m1v1=m2v2