How would the equilibrium change if the temperature were increased in so2(g) + no2(g) no(g) + so3(g) + heat?
Solved
Show answers
More tips
- F Family and Home How to Choose a Name for Your Baby?...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry Abob of mass of 0.18 kilograms is released from a height of 45 meters above the ground level. what is the value of the kinetic energy gained by the bob at the ground level?...
- C Chemistry How many alkenes have the formula c5h10? a. 4 b. 5 c. 6 d. 7?...
- C Chemistry Amixture 21.7 g nacl 3.74 g kcl and 9.76 g licl how many moles of nacl are in this mixture...
- C Chemistry In a neon tube, what occurs immediately after an atom is excited?...
- C Chemistry Me pl brainliest to correct answer...
- C Chemistry What is required in order to melt a network solid?...
- C Chemistry Restaurants offer related but differentiated products to their consumers. in the long run, new restaurants enter the market and imitate the cuisine and atmosphere of successful...
- C Chemistry Gluconeogenesis is the conversion of pyruvate to glucose. this metabolic pathway needs several cofactors to complete the conversion. complete the unbalanced equation to...
- C Chemistry :X: The image represents a(n)...
- C Chemistry If a substance is ionic, then it likely will...

Ответ:
Equilibrium change for temperature will depend on the type of reaction.
There are two types of reactions:
1. Endothermic reaction : It is a chemical reaction which absorb energy in the form of heat or light. Heat is on reactant side.
2. Exothermic reaction : It is a chemical reaction which releases energy in the form of heat or light. Heat is on product side.
Our given reaction :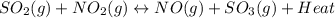 is exothermic reaction because it releases heat and heat is present on product side.
is exothermic reaction because it releases heat and heat is present on product side.
According to Le Chatelier's principle , the reaction will proceed in such a way as to counteract the temperature change.
Reason : If we increase the temperature , that means we are adding heat to the system and in our given reaction heat is present on right side, so increase in temperature will increase the heat on right side which will disturb the equilibrium and to counteract the temperature change and to re-establish the equilibrium again , the reaction will proceed to left side (Reactant side)(Reverse reaction is favored).
Note : The exothermic reaction will favor the reverse reaction , opposite the side of heat. And endothermic reaction will favor the forward reaction with increase in temperature.
Ответ:
C)sodium hydroxide
Explanation:
hope this can help you