Hydrocortisone valerate is an ingredient in hydrocortisone cream, prescribed for skin problems. its molecular formula is c26h38o6. what is the percent by mass of carbon in hydrocortisone valerate?
Solved
Show answers
More tips
- S Style and Beauty How to Tie a Tie Correctly: A Simple Guide...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry what is released when the chemical bond is broken between the second and third phosphates of an atp molecule...
- C Chemistry Please help Balancing chemical equations...
- C Chemistry Radiation sources that come from Earth itself are called radionuclides....
- C Chemistry Excess Ca(OH)2 is shaken with water to produce a saturated solution. The solution is filtered, and a 50.00 mL sample titrated with HCl requires 11.15 mL of 0.0973 M HCl to...
- C Chemistry Determine the density of a substance having a mass of 57.2 g and a volume of 33.8ml...
- C Chemistry You mix a blue and a clear liquid together and they turn a bright green. has a chemical reaction occurred? why or why not...
- C Chemistry Which of these rocks forms from lava that cools very quickly? a.schist b.gneiss c.granite d.obsidian...
- H Health What is the main problem with the following critique? “I’ve never liked horses, so I didn’t think your speech was very interesting. Besides, the way your voice sounds has always...
- H History How has population growth affected global societies during the 20th and 21st centuries...
- A Arts Which of the following ideas is further developed by Edna s inability to refuse Robert? Edna is the dominant partner in this relationship. Edna is destined to lose Robert to...

Ответ:
Answer : The percentage by mass of carbon in hydrocortisone valerate is 69.95 %.
Solution :
The molecular formula is .
.
The molar mass of Carbon = 12 g/mole
The molar mass of Hydrogen = 1 g/mole
The molar mass of Oxygen = 16 g/mole
First we have to calculate the mass of one mole of .
.
In this molecule, there are 26 carbon atom, 38 hydrogen atom and 6 oxygen atom.
Mass of 26 moles of carbon = 26 moles × 12 g/mole = 312 g
Mass of 38 moles of hydrogen = 38 moles × 1 g/mole = 38 g
Mass of 6 moles of oxygen = 6 moles × 16 g/mole = 96 g
Total mass of one mole of = 312 + 38 + 96 = 446 g
= 312 + 38 + 96 = 446 g
Now we have to calculate the percent by mass of carbon in hydrocortisone valerate.
%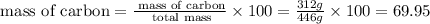 %
%
Therefore, the percentage by mass of carbon in hydrocortisone valerate is 69.95 %.
Ответ:
solid ice, when heated, turning into liquid water
Explanation: