deijiahshatia
25.03.2021 •
Chemistry
Identify the element below
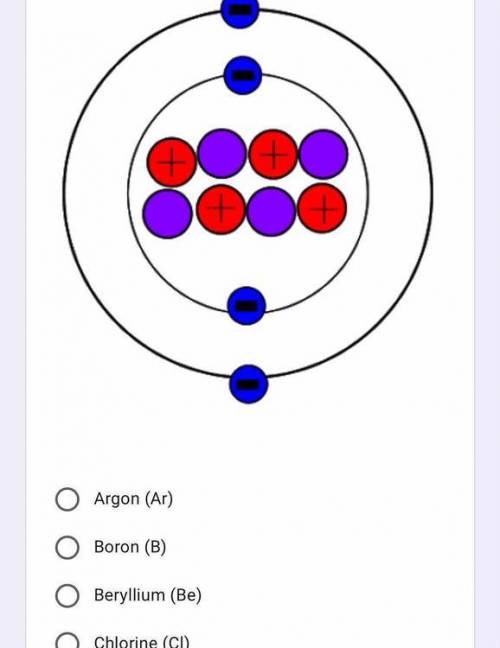
Solved
Show answers
More tips
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- M Mathematics Write an explicit rule for the recursive rule. a1=−2,an=9an−1...
- C Chemistry What makes a scientist a scientist?...
- E English Me. question is in the picture...
- A Arts Lyric game got a good alibi and my bags all packed don t u know u should never treat a girl like that cues the next ones gonna have a hammer pulled back ever girl is capable of murder...

Ответ:
Beryllium
Explanation:
Ответ:
Trial Y, because the volume of product formed per unit time is lower than Trial X.
Explanation:
The rate of reaction depends on the amount of reactants present. Now remember that the rate of reaction is either measured as the rate of disappearance of reactants or as the rate of appearance of products.
In this case, we consider the rate of reaction as the rate of appearance of products per unit time. Reading off from the graph, it is quite easy to observe that the volume of product formed per unit time for trial Y is lower than that of Trial X. Since we have earlier agreed that the rate of reaction depends on the initial concentration of reactants. It follows that the reason for the lower volume of product formed per unit time for trial Y is because of a lower initial concentration of the reactants in trial Y. Hence the answer.