jasmine2919
25.10.2019 •
Chemistry
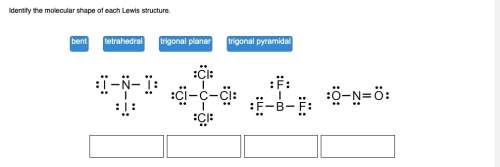
Identify the molecular shape of each lewis structure.


Solved
Show answers
More tips
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry Provide the mass number of the following isotope fe, which has 29 neutrons...
- C Chemistry 1. Holocene Rock layer rock layer name. Remember to use the Law of Superposition to determine your answers. 1.2 million years ago 2. Pleistocene Rock layer 5,000 years...
- C Chemistry Martin uses 5/8 of a gallon of paint to cover4/5 of a wall. what is the unit rate at which martin paints in walls per gallon...
- C Chemistry Consider the reaction below. hi + h2o - h3o+ + i– which is an acid-conjugate base pair? a. hi and h2o b. h2o and h3o+ c. h3o+ and i– d. hi and i–...
- C Chemistry The molar mass of SO3 is?...
- C Chemistry What is a limitation of using a chemical formula, such as C&H 208, to represent a compound? O The chemical formula does not show the types of elements that make up...
- C Chemistry Discuss: Based on the results of the experiment, which samples from the table above are likely to undergo photosynthesis?...
- C Chemistry A gas is under a pressure of 760torr and occupies volume of 525cm3. If the pressure is doubled, what volume would gas now occupy? Assume the temperature is constant?...
- C Chemistry When bismuth-214 emits a beta particle, the remaining daughter nucleus is...
- B Business What are six types of records your company is likely to need?...

Ответ:
Panel One
Trigonal Planar: This is in the shape of a triangle. It is an equilateral triangle with three equal atoms/ions at each corner of the triangle. The most well known example is a molecule with Boron at the center and the corner molecules some like Florine or iodine.
Trigonal Planar is the second Lewis Diagram from the right.
The uploaded picture is the second from the left.
==========
Tetrahedral (Second Lewis Diagram from the left) is something like a pyramid except that that the base is triangular as are each of the faces. The carbon is in the center and the chlorines (in this example) are each in a corner. If this made of just carbon (like diamond or anthracite coal) then you are talking about something that is extremely hard. It's the first upload on the left.
=====================
The Trigonal pyramid is the first Lewis Diagram on the left. It usually has an unattached pair of electrons. Nitrogen or Phosphorous are ususally the samples in the center. The nitrogen with its unused paired electrons are at the peak of blue sphere in the diagram that is uploaded second from the right.
======================
The Bent one is first lewis diagram on the right. It bends because of the unbalance (and double bonds) away from the nitrogen. Water looks something like that. Water is an example of Bent.
The upload is first on the right. These diagrams help you visulize what the various molecules look like
Summary of Lewis Diagrams.
From Left to right on the diagrams you gave me.
Trigonal Pyramidal
Tetrahedral
Trigonal Planar
Bent
Summary of uploaded diagrams.
From Left to right
Tetrahedral
Trigonal Planar
Trigonal Pyramidal
Bent
Problem Two
The very quick answer is E. Water is a bent molecule. It is flat. It has a very definite (+) end and a very definite (-) end. The marriage of a plus end with a minus is a very long lasting relationship. It is not the strongest bond in the world, but it is strong enough to create all the properties that the stem of the question has listed. [High melting and boiling points and high surface tension.
A is a repulsive condition. Likes repulse. Wrong answer.
B is wrong. We are talking about water itself, not water with something in it.
C is Wrong. Water molecules are distinct. They are a bit on the hermit side. They do not connect directly with each other.
D Wrong. There is no interchange of electrons between molecules.
Ответ:
1. VSEPR Theory
(a) NI₃
3 Bonding pairs + 1 lone pair = 4 electron groups
4 electron groups ≡ tetrahedral electron geometry
For molecular geometry, we ignore the lone pair, so the molecular shape is trigonal pyramidal (see Figure 1).
(b) CCl₄
4 bonding pairs = 4 electron groups
4 electron groups ≡ tetrahedral electron geometry
There are no lone pairs, so electron geometry = molecular geometry = tetrahedral (see Figure 2).
(c) BF₃
3 bonding pairs = 3 electron groups
3 electron groups = trigonal planar electron geometry
There are no lone pairs, so electron geometry = molecular geometry = trigonal planar (see Figure 3).
(d) NO₂ ⁻
1 single bond + 1 double bond + 1 lone pair = 3 electron groups
3 electron groups = trigonal planar electron geometry
For molecular geometry, we ignore the lone pair, so the molecular shape is bent (see Figure 4) .
===============
2. Structure and physical properties
E. The negative side of each water molecule is attracted to the positive side of nearby molecules.
Explanation:
The O-H bonds in water are highly polar, so the H atoms have a partial positive charge, and the O atoms have a partial negative charge.
The partially negative O atom in one molecule is strongly attracted to the partially positive H atom in another molecule, and vice versa.
We call these strong attractive forces hydrogen bonds.
A. Wrong. The partially positive hydrogen atoms repel each other.
B. Wrong. The polarity is a property of the bond. It is not affected by dissolved ions.
C. Wrong. The water molecules are NOT connected to each other by covalent bonds.
D. Wrong. The water molecules haven’t lost or gained electrons.
Ответ:
A
Explanation: