choiboiqg2187
25.09.2019 •
Chemistry
If 200 g of benzoyl peroxide are used with 38 kg of ptfe monomer, calculate the expected molecular weight of the polymer if (i) all the termination occurs by combination (ii) all the termination occurs by disproportionation. assume 15% efficiency for the initiator.
Solved
Show answers
More tips
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- M Mathematics Standardized test scores for all 525 students at a high school are shown below. Hide and shuffle all of the values. Take a random sample of 29 values from the population....
- M Mathematics Which of the following inequalities matches the graph? a.) 3x-2y≥4 b.) 3x-4y≤2 c.) 3x-2y≤4 d.) the correct inequality is not listed...
- H History Aprovides what is necessary to support life, not products to be sold for profit. market economy independent economy subsistence economy...
- P Physics Which best summarizes what happens to the system after heat is added? kinetic energy within the system decreases and creates mechanical energy as the piston moves upward....

Ответ:
Explanation:
The molecular weight of benzoyl peroxide is 242.23 g/mol.
Therefore, number of moles present in 200 g of benzoyl peroxide are as follows.
No. of moles =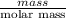
=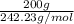
= 0.825 mol
Molecular weight of PTFE is 100 g/mol.Hence, calculate the number of moles of PTFE in 38 kg as follows.
As 1 kg = 1000 g. So, 38 kg = 38000 g.
No. of moles =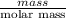
=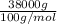
= 380 mol
As it is given that the initiator is 15% active. So, actual number of moles of benzoyl peroxide are as follows.
= 0.124 mol
For combination, 1 mol of benzoyl peroxide produces 1 chain. Therefore, degree of polymerization will be calculated as follows.
DP =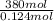
= 3065.33 mol
Average molecular weight of monomer is as follows.
= 100.05 g/mol
Therefore, calculate the expected molecular weight of polymer as follows.
= 306675.23 g/mol
Hence, for disproportion 1 mole benzoyl peroxide produces 2 chains.
DP =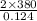
= 6129.03
Therefore, calculate the expected polymer weight as follows.
= 613209.68 g/mol
Thus, we can conclude that the the expected polymer weight is 613209.68 g/mol.
Ответ:
Step-by-step explanation:
f(4) = 42 + 5 =21, f(-10) = (-10)2 +5 = 105 or alternatively f: x → x2 + 5.
The phrase "y is a function of x" means that the value of y depends upon the value of x, so:
y can be written in terms of x (e.g. y = 3x ).
If f(x) = 3x, and y is a function of x (i.e. y = f(x) ), then the value of y when x is 4 is f(4), which is found by replacing x"s by 4"s .
If f(x) = 3x + 4, find f(5) and f(x + 1).
f(5) = 3(5) + 4 = 19
f(x + 1) = 3(x + 1) + 4 = 3x + 7