carleenespriu
14.07.2019 •
Chemistry
If 25.16 g of chlorine react with 12.99 g of manganese metal, what is the empirical formula of the compound?
Solved
Show answers
More tips
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
Answers on questions: Chemistry
- E English What is the allusion in scene 2 line 47 of romeo and juliet?...
- B Biology Which form of co2 transport accounts for the least amount of co2 transported in blood? a. as carbon monoxide in plasma b. chemically bound to hemoglobin c. as bicarbonate...
- M Mathematics During an experiment, Vanessa measured the time it took for a ball to roll down a ramp. She measured that it took 1.071 seconds. The exact time it should take based...
- E English The greek root demo means people. which word most likely comes from this root?...

Ответ:
We use the given masses of the reactants to calculate the moles of Mn and Cl. Empirical formula represents the simplest mole ratio of atoms present in a compound.
Moles of Mn =
Moles of Cl = = 0.710 mol Cl
= 0.710 mol Cl
Simplest mole ratio:
So the empirical formula is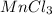
Ответ:
moderate negative correlation on edge
Step-by-step explanation: