hamadehassan
24.10.2021 •
Chemistry
In which year were the populations of light and dark colored moths the most different?
Solved
Show answers
More tips
- F Family and Home How to Properly Use a Water Level?...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry Describe the dependence of the melting point of a fatty acid upon (a) chain length and (b) unsaturation; (c) explain these dependencies in molecular terms....
- B Biology Can someone pls write this for me i’ll mark brainliest...
- C Chemistry Calculate the number of hydrogen molecules in solution prepared by mixing 45 g glucose in 36 g of water....
- M Mathematics Which of these is a correct statement?...

Ответ:
1900
Explanation:
I’m not sure but in 1900 98% of the moths were dark
Ответ:
Explanation:
Molarity is defined as the number of moles per liter of solution.
Mathematically, Molarity =
Since it is given that the molarity of a solution of 14.0 g and volume is 150 mL or 0.15 L.
Whereas number of moles =
So, molar mass of is 97.94 g/mol.
is 97.94 g/mol.
Thus, number of moles =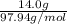
= 0.142 mol
Therefore, calculate the molarity as follows.
Molarity =
=
= 0.946 mol/L
Hence, we can conclude that molarity of the solution is 0.946 mol/L.