electrofy456
11.04.2020 •
Chemistry
Objects 1 and 2 float as shown in the beaker below. The density of the liquid in the beaker is 3.2 g/cm3. Which of the densities given below are possible values for the densities of the two objects?
Solved
Show answers
More tips
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
- H Health and Medicine How to Deal with Heat Stroke?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
Answers on questions: Chemistry
- C Chemistry The acceleration of an object depends upon...
- C Chemistry Identify the following as abiotic or biotic. a paramecium swimming in a lake...
- C Chemistry What compounds break up into ions solution?...
- C Chemistry What is the energy of an electromagnetic wave with a electromagnetic wave with a frequency of 3 * 10 Hz ?...
- C Chemistry What volume of solution must be added to 4.0 mol of NaCl to make a 1.2 M solution?...
- C Chemistry Why do animals and plants have different habitats?...
- C Chemistry Which alcohol has the strongest intermolecular force of attraction? the weakest? explain.a. 1-propanolb. 1-butanolc. n-pentaned. n-hexanee. methanolf. ethanol...
- C Chemistry An element x has a dibrom ide with the empirical formula xbr2 and a dichloride with the empirical formula xci2. the dibromide is completely converted to the dichloride...
- C Chemistry An atomic cation with a charge of +1 has the following electron configuration: 1s^2 2s^2 2p^6 3s^2 3p^5 what is the chemical symbol for the ion? how many electrons does...
- H History Ways Mao Zedongs programs could have improved or something to recommended him (Great Leap Forward) (Cultural revolution)...

Ответ:
The percent yield of the reaction is 32.34 %
Explanation:
To calculate the number of moles, we use the equation:
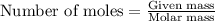 .....(1)
.....(1)
For benzoic acid:Given mass of benzoic acid = 3.6 g
Molar mass of benzoic acid = 122.12 g/mol
Putting values in equation 1, we get:
The chemical equation for the reaction of benzoic acid and methanol is:
By Stoichiometry of the reaction
1 mole of benzoic acid produces 1 mole of methyl benzoate
So, 0.0295 moles of benzoic acid will produce =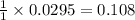 moles of methyl benzoate
moles of methyl benzoate
Now, calculating the mass of methyl benzoate from equation 1, we get:Molar mass of methyl benzoate = 136.15 g/mol
Moles of methyl benzoate = 0.0295 moles
Putting values in equation 1, we get:
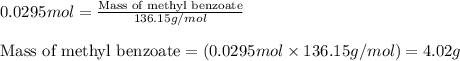
To calculate the percentage yield of methyl benzoate, we use the equation:Experimental yield of methyl benzoate = 1.3 g
Theoretical yield of methyl benzoate = 4.02 g
Putting values in above equation, we get:
Hence, the percent yield of the reaction is 32.34 %