gracethegreat1
19.10.2019 •
Chemistry
Oxygen is produced by the reaction of sodium peroxide and water.
2na2o2(s) + 2h2o(l) > o2(g) + 4naoh(aq)
a. calculate the mass of na2o2 in grams needed to form 4.80g of oxygen.
b. how many grams of naoh are produced when 4.80 g of o2 is formed?
c.when 0.48g of na2o2 is dropped in water, how many grams of o2 are formed?
Solved
Show answers
More tips
- L Leisure and Entertainment Learn How to Knit with Needles: A Simple Guide for Beginners...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
Answers on questions: Chemistry
- H History Which event created barriers in the path toward democracy in haiti?...
- G Geography the conversion of farmable or habitable land to desert, as by a change in climate or destructive land use, is known as a) transformation. b) global warming. c) desertification....
- C Chemistry When temperature drops, the number of collisions between particles increases True? False?...
- S Social Studies I need this pretty quick pls! I will give brainliest!! What happened after this event? This is a key event in The Call of the Wild. Thornton s command cracked out like a pistol-shot....

Ответ:
For a: The mass of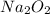 required is 23.4 grams
required is 23.4 grams
For b: The mass of NaOH required is 24 grams
For c: The mass of oxygen gas produced is 0.0984 grams.
Explanation:
To calculate the number of moles, we use the equation:
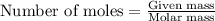 .....(2)
.....(2)
For oxygen gas:Given mass of oxygen gas = 4.80 g
Molar mass of oxygen gas = 32 g/mol
Putting values in equation 2, we get:
For the given chemical equation:
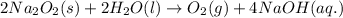
For a:By Stoichiometry of the reaction:
1 mole of oxygen gas is produced from 2 moles of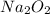
So, 0.15 moles of oxygen gas will be produced from =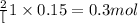 of
of 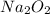
Now, calculating the mass of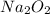 by using equation 1:
by using equation 1:
Molar mass of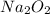 = 78 g/mol
= 78 g/mol
Moles of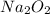 = 0.3 moles
= 0.3 moles
Putting values in equation 1, we get:
Hence, the mass of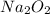 required is 23.4 grams
required is 23.4 grams
For b:By Stoichiometry of the reaction:
When 1 mole of oxygen gas is produced, then 4 moles of NaOH is also produced.
So, when 0.15 moles of oxygen gas will be produced, then =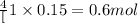 of NaOH will be produced.
of NaOH will be produced.
Now, calculating the mass of NaOH by using equation 1:
Molar mass of NaOH = 40 g/mol
Moles of NaOH = 0.6 moles
Putting values in equation 1, we get:
Hence, the mass of NaOH required is 24 grams
For c:Given mass of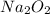 = 0.48 g
= 0.48 g
Molar mass of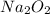 = 78 g/mol
= 78 g/mol
Putting values in equation 2, we get:
By Stoichiometry of the reaction:
2 moles of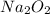 produces 1 mole of oxygen gas
produces 1 mole of oxygen gas
So,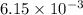 moles of
moles of 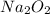 will produce =
will produce = 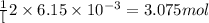 of oxygen gas
of oxygen gas
Now, calculating the mass of oxygen gas by using equation 1:
Molar mass of oxygen gas = 32 g/mol
Moles of oxygen gas =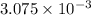 moles
moles
Putting values in equation 1, we get:
Hence, the mass of oxygen gas produced is 0.0984 grams.
Ответ:
C
Explanation: