Part A:
A mixture of carbon and sulfur has a mass of 9.0 g. Complete combustion with excess O2 gives 27.1 g of a mixture of CO2 and SO2.
Find the mass of sulfur in the original mixture.
Part B:
Calculate the mass, in grams, of a single silver atom (mAg = 107.87 amu ).
Solved
Show answers
More tips
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry For each of the following unbalanced chemical equations suppose that exactly 50.0 g of each reactant is taken. Determine which reactant is limiting, and calculate what mass of the...
- C Chemistry A balanced chemical reaction is shown below. C5H12 + 8O2 ‹ 5CO2 + 6H2O If 3 moles of C5H12 are reacted completely, how many moles of wafer are formed?...
- C Chemistry Help pls help URGENT...
- C Chemistry Aqueous solutions of compounds containing element are green. Element could be cobalt B) sulfur C) carbon D) sodium...
- C Chemistry If earths glaciers were to melt what would be the most likely effect in florida...
- C Chemistry The image at left shows three stars in the constellation Orion: Betelgeuse (A), Mintaka (B), and Rigel (C). How do the appearances of stars A, B, and C compare?...
- C Chemistry The equation of bromination of methane...
- C Chemistry Calculate the second volumes. 955 L at 58 C and 108 KPa to 76 C and 123 KPa...
- C Chemistry Write a chemical formula for this molecule....
- C Chemistry What two conversion factors would you need in order to determine the number of moles of H 2O used to run the reaction?...

Ответ:
Part A = The mass of sulfur is 6.228 grams
Part B = The mass of 1 silver atom is 1.79 * 10^-22 grams
Explanation:
Part A
Step 1: Data given
A mixture of carbon and sulfur has a mass of 9.0 g
Mass of the product = 27.1 grams
X = mass carbon
Y = mass sulfur
x + y = 9.0 grams
x = 9.0 - y
x(molar mass CO2/atomic mass C) + y(molar mass SO2/atomic mass S) = 22.6
(9 - y)*(44.01/12.01) + y(64.07/32.07)
(9-y)(3.664) + y(1.998)
32.976 - 3.664y + 1.998y = 22.6
-1.666y = -10.376
y = 6.228 = mass sulfur
x = 9.0 - 6.228 = 2.772 grams = mass C
The mass of sulfur is 6.228 grams
Part B
Calculate the mass, in grams, of a single silver atom (mAg = 107.87 amu ).
Calculate moles of 1 silver atom
Moles = 1/ 6.022*10^23
Moles = 1.66*10^-24 moles
Mass = moles * molar mass
Mass = 1.66*10 ^-24 moles *107.87
Mass = 1.79 * 10^-22 grams
The mass of 1 silver atom is 1.79 * 10^-22 grams
Ответ:
Explanation:
This is like an empirical formula question, except that you are finding the molar ratio of compounds instead of atoms.
Step 1. Gather the information in one place.
M_r: 142 18
Na₂SO₄·xH₂O(s) ⟶ Na₂SO₄(s) + xH₂O(g)
m/g: 3.22 1.42
Step 2. Calculate the mass of the water
Mass of H₂O = mass of Na₂SO₄·xH₂O – mass of Na₂SO₄
= 3.22 – 1.42 = 1.80 g
Step 3. Calculate the moles of each product
Na₂SO₄: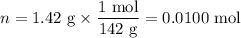
H₂O: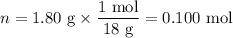
Step 4. Calculate the molar ratios