nadinealonzo6121
12.05.2021 •
Chemistry
PLEASE HELP ME If a radioactive substance has an initial mass of 50 grams
and its mass halves every 5 years, what is the number of
grams remaining after 20 years?
Solved
Show answers
More tips
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry Any guys down to start a convo...
- C Chemistry A common experiment in chemistry classes is comparing the mass and volume of different objects. The question that needs to be answered is, How are mass and volume related?...
- C Chemistry La teobromina (C7H8N4O2) de una muestra de cacao molido de 2,95 g se convirtió en la sal de plata poco soluble C7H7N4O2Ag calentando la muestra en una disolución amoniacal...
- C Chemistry A solution contains 3.75 g of a nonvolatile pure hydrocarbon in 95g acetone. The boiling points ofpure acetone and the solutions are55.95°C and 56.50°C, respectively.The...
- C Chemistry Which of the following is true about a hypothesis that is proven to be inaccurate? WILL GIVE BRAINLIEST FOR CORRECT ANSWER!! A. An inaccurate hypothesis is not valuable....
- C Chemistry One brand of extra-strength antacid tablets contains 750 mg of calcium carbonate (100 g/mol) in each tablet. stomach acid is essentially a hydrochloric acid solution....
- C Chemistry Identify the state of matter for a, b, and c. a) particles are close together in random positions with about equal kinetic energy and intermolecular forces. b) particles...
- C Chemistry Astudent weighs out 3.23 g of fe(no3)3 • ? h2o and heats it. the remaining anhydrous salt has a mass of 1.93 g. calculate the empirical formula for the hydrate....
- M Mathematics Figure ABCD is a square. Prove BD ≅ AC. Square A B C D with diagonals is shown. Statements Reasons 1. ABCD is a square 1. given 2. ∠DAB, ∠ABC, ∠BCD, and ∠CDA are right...
- B Biology What do microwaves and x-rays have in common?...

Ответ:
Explanation:
Given
Initial mass
half-life is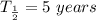
At any time the left amount is given by
Ответ:
447.63 g HClO₃
Explanation:Moles are calculated by dividing the mass of a compound by its relative formula mass.Therefore, when given the number of moles, to get the mass of the compound we multiply the number moles by the relative formula mass of the compound.Relative formula mass i given by adding the atomic masses of elements present in the compound.In this case;
Relative formula mass of HClO₃ = 84.459 g
We are given, Moles of HClO₃ = 5.3 moles
Therefore;
Mass of HClO₃ = 5.3 moles × 84.459 g/mol
= 447.6327 g
= 447.63 g (2 d.p.)
Thus, the mass of HClO₃ in 5.3 moles is 447.63 g