Please help mw with this
Solved
Show answers
More tips
- S Style and Beauty 5 Tips for Choosing the Best Ugg Boots...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- H Health and Medicine Effective Ways to Treat Colic in Infants...
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- C Cities and Countries What time does the Metro open in Moscow?...
- W Work and Career Where can you learn to be a flight attendant?...
- H Health and Medicine What are Electronic Cigarettes? The Ultimate Guide...
- C Computers and Internet Where are passwords stored in Opera?...
Answers on questions: Chemistry
- C Chemistry How can valid conclusions from the growth rate calculations be applied in a biotechnological or biomedical context ?...
- C Chemistry Under certain conditions, neon (ne) gas diffuses at a rate of 7.0 centimeters per second. under the same conditions, an unknown gas diffuses at a rate of 4.9 centimeters per...
- C Chemistry Write the quantities 3 × 10^-4 cm and 3 × 10^4 km in ordinary notation...
- C Chemistry What gas is dissolved in carbonated soda...
- C Chemistry Use your data to answer the following questions. show all of your calculations. a.what is the order for i2? (2 pts) b. what is the order for acetone? (2 pts)c.what is the order...
- C Chemistry What happens when the data in an investigation do not support the original hypothesis...
- C Chemistry When 1.00 mole of NH4NO3 dissolves in water, the enthalpy change is ΔH = + 25.7 kJ. What is the enthalpy change if 17.5 grams of NH4NO3 are dissolved...
- C Chemistry Im bored and im supposed to be doing school someone entertain me pls...
- C Chemistry Calculate ΔG ° for the following reactions at 25 ° C: a) 2Mg (s) + O2 (g) → 2MgO (s); b) 2SO2 (g) + O2 (g) → 2SO3 (g); c) 2C2H6 (g) + 7O2 (g) → 4CO2 (g) + 6H2O (l)...
- C Chemistry The car has a mass of 1000 kg and the maximum forward force on the car, produced by the engine, is 4000 N. It is claimed that the car will accelerate from 0 to 24 m/s in 6 seconds....

Ответ:
I cannot see any picture
Explanation:
Please put a picture
Ответ:
sure what's the question
Explanation:
Ответ:
Answer : The molarity of chloride anion in the solution is 0.05 mole/L
Explanation : Given,
Mass of = 0.701 g
= 0.701 g
Volume of solution = 50 ml = 0.050 L
Molarity of AgCl = 55.0 mM = 0.055 M
Molar mass of = 126.751 g/mole
= 126.751 g/mole
First we have to calculate the moles of .
.
Now we have to calculate the moles of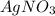 .
.
Now we have to calculate the limiting and excess reagent.
The balanced chemical reaction is,
From the balanced reaction we conclude that
As, 2 moles of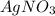 react with 1 mole of
react with 1 mole of 
So, 0.00275 moles of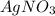 react with
react with 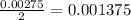 moles of
moles of 
From this we conclude that, is an excess reagent because the given moles are greater than the required moles and
is an excess reagent because the given moles are greater than the required moles and 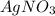 is a limiting reagent and it limits the formation of product.
is a limiting reagent and it limits the formation of product.
Now we have to calculate the moles of .
.
As, 2 moles of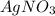 react to give 2 moles of
react to give 2 moles of 
So, 0.00275 moles of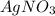 react to give 0.00275 moles of
react to give 0.00275 moles of 
Now we have to calculate the molarity of .
.
As we know that, 1 mole of AgCl in solution gives 1 mole of silver ion and 1 mole of chloride ion.
So, the molarity of chloride ion = Molarity of AgCl = 0.05 mole/L
Therefore, the molarity of chloride anion in the solution is 0.05 mole/L