ashleychantal99
21.05.2021 •
Chemistry
PLEASE HELPP!!
What is the empirical formula of the compound is C22H44O22
A. CH2O
B. C3H6O3
C. C2H4O2
D. CHO

Solved
Show answers
More tips
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry Which of these terms describes the expression of an organism’s genes?...
- H History How did the xyz affair get its name? -each letter represented the first initial of the french representatives names. -the americans referred to the french representatives...
- E English Why does macbeth accept the second and third apparitions?...
- M Mathematics YOU NEED TO HELP PLZ 4 * 10,000 + 3 * 1,000 + 2 * 100 + 7 + 5 * 1/10 / 10 ^ 3 ROUNDS TO THE NEAREST HUNDREDTH MULTIPLY THIS BY 19 SUBTRACT 43 AND TWENTY TWO HUNDREDTHS....

Ответ:
Solution :
Time (sec) Volume of NaOH (mL)
339 26.23
1242 27.80
2745 29.70
4546 3.81
Now the example of the first order kinetics w.r.t volumetric analysis is :
Here,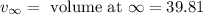
Since the interval is not constant, we take the time interval as
= 1402.3333
≈ 1402 seconds
= 0.001643 x 0.52045
= 0.00082
Therefore, the first order rate constant is k .
.