Question 1 (4 points) Using the 4 pictures below, write a lab safety rule that could go with EACH of the pictures. You should have 4 rules in total (one for each picture)

Solved
Show answers
More tips
- W Work and Career Where can you learn to be a flight attendant?...
- C Cities and Countries What time does the Metro open in Moscow?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- H Health and Medicine Effective Ways to Treat Colic in Infants...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- S Style and Beauty 5 Tips for Choosing the Best Ugg Boots...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- C Computers and Internet iPhone SMS Delivery Report: How to Set It Up...
Answers on questions: Chemistry
- C Chemistry Use the ΔHrxn values of the following reactions: 2SO2(g) + O2(g) → 2SO3(g) ΔHrxn = –196 kJ 2S(s) + 3O2(g) → 2SO3(g) ΔHrxn = –790 kJ to calculate the ΔHrxn value of this reaction:...
- M Mathematics Mr. Zhāng received a shipment that arrived in a cardboard box in the shape of a rectangular prism. The volume of the box was 110 cubic feet. The area of the base of the box was...
- M Mathematics _lAnalyzing Student WorkHI__on theSofia graphed the points 1.3,-1,0.25, and 1number line.+ + + + + + +-2 -1 0 1 2which statements about Sofia s work are true? Selectall that apply.She...
- E English MADI COME TO BESTIE LUV!! 3 ion even know- but madi come to the question...

Ответ:
Blank # 1 - keep safe distance from the burner
Blank # 2 - Place the equipments on the right place after use
Blank # 3 - perform experiments with proper lab equipment and safety tools
Blank # 4 - Don't break any test tube or lab equipment
(maybe it can be hazardous)
Ответ:
Answer :
Formal charge on C: (-1)
Formal charge on N: (0)
Net charge: (-1)
Explanation :
First we have to determine the Lewis-dot structure of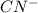 .
.
Lewis-dot structure : It shows the bonding between the atoms of a molecule and it also shows the unpaired electrons present in the molecule.
In the Lewis-dot structure the valance electrons are shown by 'dot'.
The given molecule is,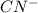
As we know that carbon has '4' valence electrons and nitrogen has '5' valence electrons.
Therefore, the total number of valence electrons in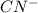 = 4 + 5 + 1= 10
= 4 + 5 + 1= 10
According to Lewis-dot structure, there are 6 number of bonding electrons and 4 number of non-bonding electrons.
Now we have to determine the formal charge for each atom.
Formula for formal charge :
Net charge = -1 + 0 = -1