alexabdercmur
21.04.2020 •
Chemistry
QUESTION 5
A balloon has been filled to a volume of 2.90 L with 0.150 mol of helium gas. If 0.0450 mol of additional helium is added to the balloon while
the temperature and pressure are held constant, what is the new volume of the balloon?
Hint: Avogadro's law is V7/n1=V2/n2
A. 3.95 L
B. 3.77 L
C. 6.54 L
D. 5.45 L
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- S Style and Beauty How to Sew Harem Pants?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry Carbon dioxide is usually formed when gasoline is burned. If 3.00 L of CO2 is produced at a temperature of 100 °C, what is the temperature of the gas if the volume...
- C Chemistry Use the graph to answer these questions. For numerical answers, use correct significant digits. Letter shows the overall enthalpy of reaction which is equal to kJ....
- C Chemistry A student prepares a aqueous solution of acetic acid . Calculate the fraction of acetic acid that is in the dissociated form in his solution. Express your answer...
- C Chemistry Mark all statements that are true in regards to mole ratios. A mole ratio is the central step in all stoichiometry problems Mole ratios can be established from a...
- C Chemistry A) 2.82 L B) 0.355 L C) 2.88 L D) 0.347 L...
- C Chemistry How many energy levels will argon have?...
- C Chemistry Calculate the average atomic mass of chlorine if 75.77% of its atoms have a mass of 34.97 amu and 24.23% of its atoms have a mass of 36.96 amu....
- C Chemistry Why can t the position of an electron be determined with certainty?...
- C Chemistry Marble or small speaker made of stone is found to have a mass of 3.05 g to find its volume it is placed in a graduated cylinder that originally contained 10.00 ml...
- C Chemistry The half-life of a radioisotope is the amount of time it takes for a. half the sample to decay. b. all the sample to decay. c. the age of an artifact to be calculated....

Ответ:
B. 3.77 L is the new volume occupied by the gas.
Explanation:
As per Avogadro's law, which states that if the pressure and temperature held constant, then an equal volume of the gases will occupy an equal number of molecules. It can be written as,
Here, V1, volume of the helium gas = 2.9 L
V2, volume of the additional helium gas in the balloon = ?
n1, moles of helium gas = 0.150 mol
n2, number of moles of additional helium gas = 0.150 + 0.0450 = 0.195 mol
We have to rearrange the equation for V2 as,
V2 =
Now Plugin the values as,
V2 =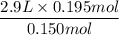
= 3.77 L
So the new volume of the balloon is 3.77 L.
Ответ:
Step-by-step explanation:
r u in public skl or priv?