apodoltsev2021
06.10.2019 •
Chemistry
Questions 3-6 refer to rhe solutions below:
(a)100ml of 1.0 m ch3cooh mixed with 100ml of 1.0 m ch3coona
(b)100ml of 1.0 m hbr mixed with 100ml of 1.0 m kbr
(c)100ml of 1.0 m hi mixed with 100ml of 1.0 m naoh
(d)100ml of 1.0 m nh4cl mixed with 100ml of 1.0 m nh3
(e)100ml of 1.0 m naoh mixed with 100ml of 1.0 m ch3cooh
3.) which solution will produce a buffer with a ph < 6.5
4.) which solution will produce a buffer with a ph > 7.5
5.) which solutiom will have a ph of about 7
6.) which solution will be the most acidic
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- S Style and Beauty How to Sew Harem Pants?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry If an atom loses 2 electrons, what is the charge on the resulting ion what is an example...
- C Chemistry Labels, 2) What type of seismic wave will reach a recording station 500 miles away from the epicenter first? Explain why,...
- H Health The scene is safe, you ve turned on the AED and it prompts you to attach the pads. Choose the best answer for correct both pads placement on a 6 year old child cardiac...
- B Business Oakville Trucking just signed a $5.0 million contract. The contract calls for a payment of $1.25 million today, $1.75 million one year from today, and $2.0 million...
- P Physics Set up the hamiltonian and hamilton’s equations for a projectile of mass m, moving in a vertical plane and subject to gravity but no air resistance. Use as your coordinates...
- E English At key junctures, in the essay, gladwell makes sharp personal comments. In fact his ending viva la revolucion could be read as downright sarcastic what others do...
- B Business Management of Mittel Rhein AG of Köln, Germany, would like to reduce the amount of time between when a customer places an order and when the order is shipped. For...
- C Chemistry entify the hybridization state and geometry of each carbon atom in the following compound:Write the carbon atom(s) in numerical order as a comma separated list (e.g....
- P Physics Identify the composition of the unknown objects based on their densities. If there are several possibilities (due to the percent uncertainty) state which possibility...
- B Business Make or Buy A company manufactures various-sized plastic bottles for its medicinal product. The manufacturing cost for small bottles is $75 per unit (100 bottles),...

Ответ:
Under room temperature where :
:
3.) (A), (B), and (E).
4.) (D).
5.) (B).
ExplanationWhat makes a buffer solution? For a solution to be a buffer, it needs to contain large amounts of a weak acid and its conjugate base ion. Alternatively, the solution may contain large amounts of a weak base and its conjugate acid ion.
Not every one of the five solutions is a buffer solution.
(A)Ethanoic acid CH₃COOH (a.k.a. acetic acid) is a weak acid. pKa = 4.756. CH₃COONa is a salt. It dissolves to produce CH₃COO⁻, which is the conjugate base ion of CH₃COOH. The solution in (A) contains equal number of CH₃COOH and CH₃COO⁻, both at 1.0 M.
Refer to the Henderson-Hasselbalch equation for buffers of weak acids.
The pH of the solution in (A) will be the same as the pKa of CH₃COOH. pH = 4.746.
(B)Consider the hydrogen halides:
HF: weak acid.HCl: strong acid.HBr: strong acid.The radius of halogen atoms increases down the group, and hydrogen-halogen bond becomes weaker. It becomes easier for water to break those bonds. As a result, the strength of hydrogen halides increases down the group. HF is the only weak acid among the common hydrogen halides.
Mixing HBr and KBr at equal ratio will be similar to mixing HCl and KCl at the same ratio. All HBr in the solution breaks down into H⁺ and Br⁻. The pH of the solution will depend only on the concentration of HBr.
![\text{pH} = -\log{[\text{H}^{+}] = -\log{0.50} \approx {\bf 0.30}](/tpl/images/0294/2613/3101f.png) .
.
(C)Similarly to HCl and HBr, HI is also a strong acid. Mixing HI and NaOH at equal ratio will produce a solution of NaI, which is similar to NaCl. The final solution will be neutral. pH = 7 if pKw = 14.
(D)NH₃ is a weak base. NH₄Cl dissolves completely to produce NH₄⁺ and Cl⁻. NH₄⁺ is the conjugate acid of NH₃. The final solution will contain an equal number of NH₃ and NH₄⁺. pKb = 4.75 for ammonia NH₃.
Apply the Henderson-Hasselbalch equation for buffers of weak bases:
Note that what this equation gives for buffers of weak bases is the pOH of the solution. pH = pKw - pOH. Assume that pKw = 14. pH = 14 - 4.75 = 9.25.
(E)The solution in (E) will contain about 1.0 M of CH₃COOH. The volume of the solution will be 200 mL.
There's nearly no conjugate base of CH₃COOH. As a result, the solution will not be a buffer, and the Henderson-Hasselbalch Equation will not apply. Refer to an ICE table:
The value of pKa is large. Ka will be small. the value of will be much smaller than
will be much smaller than  such that
such that 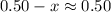 .
.
The pKa of a weak acid is the same as pKw divided by the pKb of its conjugate base.
Ответ: