melissapulido198
03.06.2021 •
Chemistry
Rank these transition metal ions in order of decreasing number of unpaired electrons.
a. Fe^3
b. Mn^4+
c. V3+
d. Ni^2+
e. Cu^+
Solved
Show answers
More tips
- A Animals and plants Money Tree Care Secrets: How to Keep Your Plant Thriving...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- P Philosophy How to Develop Extrasensory Abilities?...
- S Style and Beauty Don t Sacrifice Your Brows: How to Properly Pluck Stubborn Hairs...
Answers on questions: Chemistry
- C Chemistry How do heatwaves show physical and chemical changes...
- C Chemistry Black cloth will appear red through a red glass. True or False...
- C Chemistry For selenium trioxide, answer the following: e) determine formal charges to all atoms f) does this structure that you drew have resonance? If so, draw all possible resonance...
- C Chemistry Can someone help me please...
- C Chemistry Can someone help me please...
- C Chemistry How many milliliters of a stock solution of 6.80 MM HNO3HNO3 would you have to use to prepare 0.110 LL of 0.470 MM HNO3HNO3 ?...
- B Business Ihave a few questionsoperate systems for gathering organizing and distributing information. 1. financial managers 2. human resources managers 3. information managers 4. controllers...
- H History How did the weather affect the building of the panama canal? the hot, dry weather made many of the machines break down. the cool, rainy weather construction move quickly. the...
- M Mathematics Abaseball team has home games on friday and saturday. the two games together earn 3979.00 for the team. fridays game generates 781.00 less then saturdays game. how much money...
- M Mathematics Adog chased after a dandelion for 0.5 km, stopped to take a rest, and continued to chase after the dandelion for 750 m. how many meters did the dog travel in total?...

Ответ:
The given transition metal ions in order of decreasing number of unpaired electrons are as follows.
Explanation:
In atomic orbitals, the distribution of electrons of an atom is called electronic configuration.
The electronic configuration in terms of noble gases for the given elements are as follows.
Atomic number of Fe is 26.So, there is only 1 unpaired electron present in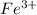 .
.
Atomic number of Mn is 25.So, there are only 3 unpaired electrons present in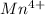 .
.
Atomic number of V is 23.So, there are only 2 unpaired electrons present in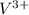 .
.
Atomic number of Ni is 28.So, there will be 2 unpaired electrons present in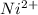 .
.
Atomic number of Cu is 29.So, there is no unpaired electron present in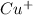 .
.
Therefore, given transition metal ions in order of decreasing number of unpaired electrons are as follows.
Thus, we can conclude that given transition metal ions in order of decreasing number of unpaired electrons are as follows.
Ответ:
Answer : The IUPAC name of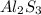 is aluminum sulfide.
is aluminum sulfide.
Explanation :
For formation of a neutral ionic compound, the charges on cation and anion must be balanced.
The cation is formed by loss of electrons by metals and anions are formed by gain of electrons by non metals.
Ionic compound : It is a type of compound that is made up of the ions of the two different elements in which one element is a metal and another element is a non-metal.
The nomenclature of ionic compounds is given by:
Positive is written first.The negative ion is written next and a suffix is added at the end of the negative ion. The suffix written is '-ide'.The given compound is,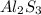
Here aluminum is having an oxidation state of (+3) called as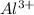 cation and sulfur
cation and sulfur 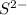 is an anion with oxidation state of (-2). Thus, they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
is an anion with oxidation state of (-2). Thus, they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral 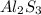 called as aluminum sulfide.
called as aluminum sulfide.
Hence, the IUPAC name of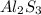 is aluminum sulfide.
is aluminum sulfide.