poreally6456
22.08.2019 •
Chemistry
Sample a is a brand of supplement that contains iron in the form of 324mg ferrous gluconate c12h24feo14 per tablet. what is the amount of elemental iron in one table of sample a?
Solved
Show answers
More tips
- H Health and Medicine How Many Ribs Do Humans Have?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- W Work and Career Мерчендайзинг – все, что нужно знать...
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
Answers on questions: Chemistry
- A Arts Trenton has a new mp3 player. he loves that it s so small and sleek. the little screen on the front tells him what song is playing and even what time it is. however, he;s having trouble...
- W World Languages Friend me just for fun got nothing else to do...
- E English The knights at the round table express their solidarity by wearing a green girdle as gawain does. why does gawain wear the green girdle?...
- A Arts Alex wants to paint kne side of his skateboard ramp with glow in the dark paint but he needs to know how much area he is painting calculate the area of the isosceles trapezoid....
- M Mathematics What is the value of x?...

Ответ:
Amount of elemental iron in sample A is 0.0405g.
Explanation:
Molar mass of C = 12 g/mol
Molar mass of H = 1 g/mol
Molar mass of Fe = 56 g/mol
Molar mass of O = 16 g/mol
So, molar mass of ferrous gluconate =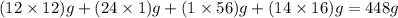
So number of mole of ferrous gluconate in 324 mg =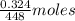
(number of moles =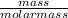 )
)
As 1 mol of ferrous gluconate contains 1 mol of Fe therefore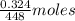 of ferrous gluconate contain
of ferrous gluconate contain 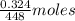 of Fe.
of Fe.
So amount of elemental iron in sample A =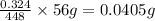
Ответ: