louiecampos
26.08.2020 •
Chemistry
Sea la reacción de síntesis de la urea: 2 NH3 + CO2 → (NH2)2CO + H2O Si tenemos 500 gramos de NH3 y 1000 gramos de CO2 calcular cuál es el reactivo limitante y la cantidad de urea producida.
Solved
Show answers
More tips
- F Family and Home How to Properly Use a Water Level?...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry What is the percent yield of water from the combustion of propane (C3H8), if my theoretical yield is 8.17g of water and my actual yield was 6.13 g water? A)Answer...
- C Chemistry How many significant figures are in the number 420,000 mi?...
- C Chemistry Water beads up on waxy surfaces because of a degree of adhesion with the surface? high low...
- C Chemistry What kind of weather does a cold front usually bring? a. warm c. sunny b. stormy d. windy select the best answer from the choices provided a b c d...
- C Chemistry Which of the following statements supports the concept that organisms are still evolving? the increasing population of humans. the decreasing number of fossils found...
- C Chemistry Earths rotation takes about whereas its revolution takes about a) one day; one year b) one year; one day c) one month; one year d) one day ; one elipse...
- C Chemistry Need with my dog! my dog can t walk or even stand when he try s to he just falls, he can move his head and he tries to get up, just yesterday he was ok, he was playing,...
- C Chemistry At 25 c, 1.00 mole of O2 gas was found to occupy a volume of 12.5L at a pressure of 198 kPa. What is the value of the gas constant in L • kPa / mol • k?...
- C Chemistry A 150 mL sample of gas is collected at 1.20 atm and 25°C. What volume does the gas have at 1.50 atm and 20.0°C?...
- C Chemistry During lab, you learned about exothermic and endothermic heats of solution. Which compounds below could be used for a cold pack? (Select all that apply.) a. LiCl...

Ответ:
El reactivo limite es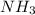 y la cantidad de urea producida es 881.58 g
y la cantidad de urea producida es 881.58 g
Explanation:
Podemos empezar con la reacción:
Tenemos 2 átomos de nitrógeno en ambos lados, 1 átomo de carbono en ambos lados, 2 átomos de oxigeno en ambos lados y 6 átomos de hidrógeno en ambos lados. Por lo tanto la reacción esta balanceada.
Ahora, para hallar el reactivo limite necesitamos seguir los siguientes pasos:
1) Convertir a moles usando la masa molecular de cada compuesto ( 17 g/mol para el amoniaco y 28 g/mol para el dióxido de carbono).
2) Usando el la relación molar entre cada compuesto con la urea podemos calcular las moles de urea ( para el amoniaco la relación es 2:1 y para el dióxido de carbono la relación es 1:1).
3) Finalmente, usando la masa molar de la urea (60 g/mol) podemos calcular la masa de urea.
El elemento que tiene la menor cantidad de urea se da en el calculo del amoniaco. Por lo tanto, el reactivo limite es el amoniaco y la cantidad de urea producida es 881.58 g
Espero que sea de ayuda!
Ответ:
False
complete combustion produces carbon dioxide + water