Tear gas has the composition 40.25% carbon, 6.19% hydrogen, 8.94% oxygen, 44.62% bromine. what is the empirical formula of this compound?
Solved
Show answers
More tips
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- B Biology The kinetic data is listed below in the absence and in the presence of an inhibitor: 1/[s] mm 1/ vo (s/mm) (w/o i) 1/ vo(s/mm) (w / x) 1.5 13.75 27.5 2.0 15 30 3.0 17.5 35...
- W World Languages 1) Esses textos têm em comum o fato de * 1 ponto A) serem textos informativos. Ambos os textos estão centralizados em fatos/acontecimentos. B) serem textos argumentativos....
- M Mathematics A right triangle has one angle that measures 24°. What is the measure of the other acute angle?...
- M Mathematics The change of base formula is used to calculate a wider range of logarithms. true false...
- B Biology The average age at which an infant can sit unattended is approximately months. a. two to three b. eight to nine c. three to five d. five to seven...

Ответ:
Empirical formula will be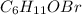
Explanation: Assuming that the total mass of the given tear gas = 100g
Mass of each element will be equal to the percentages of each element given
Mass of Carbon = 40.25g
Mass of Hydrogen = 6.19g
Mass of Oxygen = 8.94g
Mass of Bromine = 44.62g
Now, converting mass of each element into their moles using the formula
Dividing moles of each element with the smallest number of moles calculated, we get the mole ratio of each element.
The mole ratio of each element is written in the subscript of each element in the empirical formula.
Empirical Formula :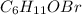
Ответ:
Hornblende
Explanation:
I'm not sure if this is it but i think it is