hannahdrumsey
10.03.2020 •
Chemistry
Ten kilograms of R-134a fill a 1.595-m3 weighted piston-cylinder device at a temperature of −26.4°C. The container is now heated until the temperature is 100°C. Determine the final volume of the R-134a.
Solved
Show answers
More tips
- S Science and Technology How to Restore the Diamond Shine to Your Tarnished Silverware...
- C Computers and Internet Boost your processor performance with these easy tips...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
Answers on questions: Chemistry
- C Chemistry How many atoms are in NaHCO3? And please explain how u got the answer....
- C Chemistry Pure phosgene gas (COCl2), 0.0340 mol, was placed in a 1.50−L container. It was heated to 700.0 K, and at equilibrium, the pressure of CO was found to be 0.509 atm. Calculate...
- C Chemistry The stress-strain curve for a brass alloy is provided below. The inset is a magnified view of the elastic regime of this stress-strain response. What is the approximate...
- C Chemistry If Kp For This Reaction Is 0.071 At 100 °C (when The Partial Pressures Are Measured In Atmospheres), What Is The Total Pressure In The Flask At Equilibrium? Total Pressure...
- C Chemistry The reaction of methyl iodide with sodium azide, NaN3, proceeds by an SN2 mechanism. What is the effect of doubling the concentration of NaN3 on the rate of the reaction?...
- C Chemistry 21. How many Co, and ATP molecules are formed during one complete turn of tricarboxylic acid cycle (Krebs cycle)?a. 200, and IATPb. 200, and 2ATPc. 200, and 16ATPd. 2002...
- B Business Adifference between supermarkets and convenience stores is that unlike supermarkets, convenience stores...
- E English One advantage of using dialogue in a narrative essay is to 1. highlight the essay’s topic. 2. define the essay’s purpose. 3. show who the essay’s characters are. 4. determine...
- E English fill in the blankk ⦁ the battle of philippi started after the assassination of ⦁ the battle of philippi took place in the year ⦁ cassius built a to stop antony from breaking...
- H History How did mesoamerican and mesopotamian farmers differ in their domestication of animals during the neolithic revolution? a. mesoamericans began domesticating animals about...

Ответ:
Explanation:
The given data is as follows.
Mass of refrigerant, m = 10 kg
Volume of the refrigerant, V = 1.595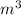
Formula for specific volume of the refrigerant is as follows.
v =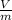
= 0.1595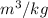
So, at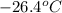 specific volume will be within
specific volume will be within 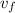 and
and 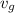 and pressure is constant.
and pressure is constant.
The fluid will be in super-heated state at temperature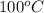 and at T =
and at T = 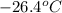 pressure 1 bar = 0.1 MPa.
pressure 1 bar = 0.1 MPa.
According to super-heated tables, the specific volume is v = 0.30138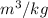 .
.
Hence, the final volume will be calculated as follows.
=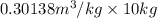
= 3.0138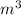
Thus, we can conclude that final volume of the R-134a is 3.0138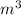 .
.
Ответ:
4x + 72 + 2x - 12 = 180
6x + 60 = 180
6x = 180 -60
6x = 120
x = 120/6
x = 20