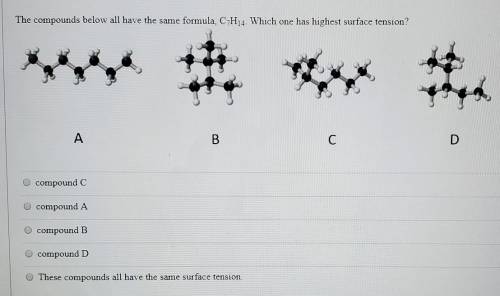
The compounds below all have the same formula, C7H14. Which has the highest surface tension?
Solved
Show answers
More tips
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- M Mathematics Rewards will be given properly which of the following numbers is irrational? a. 625.49 b. 625.89 c. v 90 d. v 900 ( a. the 49 is continual )...
- E English What crime did Kim Ima s father commit to be put in a World War II American concentration camp?...
- M Mathematics Question 1: solve for the missing length and the other two angles in the triangle below. part i: use the law of cosines to find the missing third side. part ii: use either...
- E English Which sentence shows correct subject-verb agreement? There’s a good answer to that question. There’re a good answer to that question...
- C Chemistry Willa holds a pan over a campfire, as shown in the figure below. She feels warmth from the fire. Most of the warmth Willa feels comes from the process represented by letter...

Ответ:
6.83x10⁻⁴M is the initial concentration of the Cu²⁺
Explanation:
The concentration is directly proportional to the signal (Beer-Lambert law).
The increase in concentration produce an increase in signal of:
45.1 units - 25.2 units = 19.9 units.
The increase in concentration is of:
0.500mL = 5x10⁻⁴L * (0.0275mol / L) = 1.375x10⁻⁵moles / 0.0255L = 5.39x10⁻⁴M
The conversion factor is 5.39x10⁻⁴M = 19.9 units
That means the initial concentration that produce a signal of 25.2 units is:
25.2 units * (5.39x10⁻⁴M / 19.9 units) =
6.83x10⁻⁴M is the initial concentration of the Cu²⁺