sakshimehta
24.09.2019 •
Chemistry
The reaction is as follows: ch4 + 202 + co2 + 2h2o if we have 71 kg/hr of ch4 reacting with 67 kg/hr of 02, at what rate co2 will be generated in kg/hr? molecular weight: c-12 kg/kmol h-1 kg/kmol 0 - 16 kg/kmol select one: o a. 46.062 o b. 195.250 o o o c. 1.047 d. 92.125 e. 24.364
Solved
Show answers
More tips
- F Food and Cooking What Does a Godmother Give?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- W Work and Career Мерчендайзинг – все, что нужно знать...
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
Answers on questions: Chemistry
- C Chemistry What is the total mass of a mixture of 3.50x10^22 formula units na2so4, 0.500 mol h2o, and 7.23 g agcl?...
- C Chemistry 1. Sweet grass gardens specialize in growing native plant species. In what ways are native plants particularly significant for sex nations people? How many species does the nursery...
- C Chemistry What is correct for 10g of caco3....
- C Chemistry Determine the volume (L) of nitrogen monoxide gas that is created at STP when 32.2 g of solid copper reacts with excess nitric acid. 3Cu(s) + 8HNO3(aq) — 3Cu(NO3)2 (aq) + 4H2O(1) +...
- C Chemistry To a 0.444 g sample of H2O, 23.4 J of heat are added. What is its change in temperature? The heat capacity of H2O is in Table 7.1 Specific Heats of Various Substances ....
- C Chemistry How many moles is 784.616 g of H2SO4?...
- C Chemistry Please help me with this chemistry work...
- C Chemistry A negatively charged rod is placed near to neutral metals fears, as shown below. which statement describes the charging method shown? a: the spheres are charged through friction b:...
- C Chemistry Help for Number 5 please....
- C Chemistry Rank the following gases from most to least ideal in terms of the van der Waals coefficient b: CO2, SF6, O2, H2, He, CH4, Rn. Explain the reasoning for your ranking...

Ответ:
The answers is the option a: 46.062
Explanation:
First, you must determine the limiting reagent, that is the reagent that is consumed first during the reaction.
So, in first place, you must determine the molecular weight of the molecules CH4, O2, CO2 and H20. You know that
C: 12 kg/kmol H: 1 kg/kmol 0: 16 kg/kmolSo, to determine the mass of a molecule, you must multiply the individual masses of each atom by the amount present in the molecule. This would be:
CH4: 12 kg/kmol + 4* 1 kg/kmol= 16 kg/kmol because you have 1 C and 4 H in the CH4.In the same way, you can determinate the mass of all reagents and products involved in the reaction.
O2: 2*16 kg/kmol=32 kg/kmolCO2: 12 kg/kmol+2*16 kg/kmol= 44 kg/kmolH2O: 2*1 kg/kmol + 16 kg/kmol= 18 kg/kmolNow you can apply stoichiometry to determine the limiting reagent using these numbers and observing how many molecules react.
On one side, it is known that, by stoichiometry, 1 mol of CH4 and 2 moles of O2 react. This means that 16 kg/mol of CH4 and 64 kg/kmol ( 2moles* 32 kg/kmol) of O2 react
And it is known that there are 71 kg/hr of CH4 reacting with 67 kg/hr of 02
So, using the stoichiometric information (16 kg/mol of CH4 and 64 kg/kmol of O2), 71 kg/hr of CH4 and The Rule of Three, you can determine the limiting reagent:
16 kg/kmol CH4 ⇒ 64 kg/kmol O2 (stoichiometry)
71 kg/kmol CH4 ⇒ x
So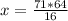
x=284 kg/kmol
This means that to react 71 kg/kmol of CH4, 284 kg/kmol of O2 are needed. But you only have 67 kg/mol that can react. That is why O2 is the limiting reagent, because it is consumed first.
Now, you can calculate the rate of CO2 that is generated, using the data of the amount of limiting reagent and stoichiometry. This is:
64 kg/kmol O2 ⇒ 44 kg/kmol CO2 (stoichiometry)
67 kg/kmol O2 ⇒ x
So
x=46.0625 kg/kmol
This means that 46.0625 kg/kmol of CO2 are generated.
Ответ:
A number of factors contributed to the rise of absolute monarchies in Europe in the seventeenth and eighteenth centuries. In the first place the disorder, anarchy and confusion which prevailed in Europe during the medieval period greatly contributed to the growth of the absolute monarchies. Hope This Helped!