senceepoai22
03.08.2021 •
Chemistry
The solubility product constant of calcium sulfate, CaSO4, is 7.10×10−57.10×10−5. Its molar mass is 136.1 g/mol. How many grams of calcium sulfate can dissolve in 75.5 L of pure water
Solved
Show answers
More tips
- C Computers and Internet 3D Glasses! What is this thing?...
- C Computers and Internet How to insert videos into LiveJournal?...
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- P Physics What is displacement? ik what it is i am just making this just bc...
- M Mathematics PLEASE HELP! PLEASE SHOW WORK Use the following expression to answer this three part question: f(x) = 2x2 + 4x − 6 Part A: What are the x-intercepts of the graph of f(x)? Show your...
- H History Where did General Robert E. Lee graduate from? Westpoint Academy Harvard O FGCU...
- B Business In Capital market there are issues of securities like a.Sharesb.Treasury billsc.Promissory notesd.All the options are wrong...

Ответ:
2)
Explanation: When an acid reacts with the base then it undergoes neutralization to form a salt and water.
1) : is an example of single replacement reaction as Mg replaces hydrogen from its salt solution.
: is an example of single replacement reaction as Mg replaces hydrogen from its salt solution.
2)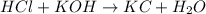 : is an example of neutralization where acid and base reacts to give salt and water.
: is an example of neutralization where acid and base reacts to give salt and water.
3) : is an example of double displacement reaction in which exchange of ions take place.
: is an example of double displacement reaction in which exchange of ions take place.
4)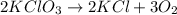 : is an example of decomposition where a single reactant decomposes to give two or more products.
: is an example of decomposition where a single reactant decomposes to give two or more products.