trashellyoung
26.10.2019 •
Chemistry
The temperature of a gas is raised from 10 to 20 degree celsius. will the volume double the pressure if mass and pressure do not change? why or why not? show calculations
Solved
Show answers
More tips
- H Health and Medicine How Many Ribs Do Humans Have?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- W Work and Career Мерчендайзинг – все, что нужно знать...
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
Answers on questions: Chemistry
- H History HUR Which are criticisms of NAFTA? Check all that apply. It has created an increase in the value of goods and services. It has caused income inequality to increase. It has led to an...
- B Biology From may to september, the worst time of day for pollution is during what hours?...
- B Business An annual reporting period consisting of any twelve consecutive months is known as:.1. Calendar year2. Natural business year3. Interim financial period 4. Fiscal year5. Seasonal year...
- M Mathematics Asurveillance camera is located on a lamp post above a parking lot. the equation of the viewing area on an overhead grid is (x – 9)2 + (y + 3)2 = 49. determine the distance from the...

Ответ:
There will be No change in volume.
Explanation:
Ideal Gas Equation is used to solve this :
PV=nRT;
P is pressure in Pa,
V is volume in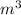 ,
,
n is number of moles of gas;
R is the universal gas constant, 8.31 J/K mol
T is temperature in Kelvin
when mass is said to be fixed, the number of moles will also be fixed, too. So combining both the constant term n and R which gives
PV=kT
where k is constant.
V=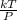
So the equation represents that, Volume V will remains unchanged when pressure and temperature are both doubled, therefore the numerator term T and the denominator term P are both gets affected by multiplication of 2, hence by cancellations:
V =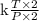 =
= 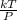
Ответ: