AdhoraM11571
14.01.2021 •
Chemistry
Uranium-238 undergoes radioactive decay according to the incomplete below. 238
92
U= 4
2 He + X.
What is the decay product represented by X?
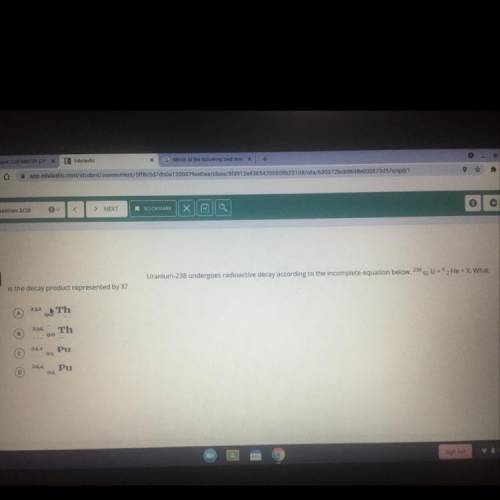
Solved
Show answers
More tips
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry The comparison to an invisible spring is pretty accurate. What kind of forces do you think are in this spring between molecules? Or, what kind of force do you think attracts...
- C Chemistry Why do sheep need grass to survive...
- C Chemistry Pls help on these two questions I will give brainlist...
- C Chemistry Why does an ice cube melt when you hold it in your hand? Your hand transfers temperature to the ice cube. Heat from the ice cube is transferred to your hand. Heat is transferred...
- C Chemistry Which statement about Niels Bohr’s atomic model is true...
- C Chemistry 1. Displacement vectors of 10 m west and 14 m west make a resultant vector that is O A 14 m west OB 24 m west OC 10 m west OD 6 m west...
- C Chemistry How many carbon atoms react in this equation? 2C4H10 + 13O2 à 8CO2 + 10H2O A) 2 B) 4 C) 6 D) 8...
- C Chemistry Can the carona virus can be cured...
- C Chemistry Two solutions are combined in a beaker. One solution contains 500.0 g of potassium phosphate and the other contains 500.0 g of calcium nitrate. A double-displacement reaction...
- S Social Studies The national conversation is responsible for......

Ответ:
The decay product represented by X : Thorium (Th) : ²³⁴₉₀Th
Further explanationGiven
Decay reaction
²³⁸₉₂U ⇒ ⁴₂He + X
Required
The decay product
Solution
Radioactivity is the process of unstable isotopes to stable isotopes by decay, by emitting certain particles,
alpha α particles ₂He⁴ beta β ₋₁e⁰ particles gamma particles ₀γ⁰ positron particles ₁e⁰ neutron ₀n¹In a decay reaction, the sum of the mass number and atomic number of the elements in the reactants and products must be the same
So the mass number of the element X = 238 - 4 = 234
Atomic number of the element X = 92 - 2 = 90
If we look at the periodic system, then the element with atomic number 90 is Thorium (Th)
Ответ: